Air Scrubber

An air scrubber is a purification system designed to remove particulate matter from the air. It achieves this by using moisture or by cooling and filtering the airstream as it passes through the scrubber.
Please fill out the following form to submit a Request for Quote to any of the following companies listed on
This article will take an in-depth look at air pollution control equipment.
You will learn about:

The term "air pollution control equipment" refers to advanced systems used to curb the discharge of various solid and gaseous pollutants into the atmosphere, primarily originating from industrial emissions via exhaust stacks or chimneys. These systems are classified into two main categories: those that target acidic gas emissions and those designed to manage particulate matter emissions effectively.

Air pollution control technologies primarily function through three core methods:
Considering that operational conditions and environments vary substantially across different facilities, choosing suitable monitoring equipment or methodologies involves more complexities than merely comparing performance metrics and costs. Each facility and installation has specific monitoring needs, influenced by several critical factors.
These factors collectively assist in acquiring the monitoring equipment best suited for specific operational requirements.
The Industrial Revolution marked a pivotal shift in manufacturing and human activity, but one major consequence was a significant rise in air pollution stemming from the combustion of fossil fuels in industrial processes. As factories, power plants, and other industrial facilities proliferated, excessive exploitation of natural resources—including wood, coal, water, and land—led to a dramatic increase in emissions of harmful pollutants such as sulfur dioxide (SO2), nitrogen oxides (NOx), carbon monoxide (CO), and particulate matter (PM).
By the mid-20th century, the serious health and environmental impacts of unchecked industrialization and poor air quality became increasingly evident around the world. For example, in 1948, severe smog in Donora, Pennsylvania, resulted in the deaths of 20 people and illnesses in more than 7,000 others—largely due to emissions from local zinc works and steel mills. In 1952, London experienced "The Great Smog," a dense, choking fog formed when sulfur dioxide, particulate matter, and coal-burning gases combined with weather conditions. This catastrophic pollution event resulted in the deaths of 12,000 people and caused respiratory distress for countless more individuals and animals over a five-day period. In 1984, Bhopal, India, became the site of another infamous air pollution disaster; a gas leak at the Union Carbide chemical plant released toxic methyl isocyanate into the air, injuring or sickening between 150,000 and 600,000 residents and killing nearly 4,000 workers.
These tragedies, along with increasing scientific evidence linking air pollution to diseases such as asthma, lung cancer, and cardiovascular conditions, prompted governments worldwide to introduce various clean air regulations and environmental protection standards. For instance, the UK government responded with the first Clean Air Act in 1956, prohibiting coal burning in cities and establishing emission limits. The United States followed with the Clean Air Act of 1970, fundamentally changing the regulatory landscape for industrial air emissions and setting strict national air quality standards.
Following the implementation of clean air laws, many industries adopted advanced air pollution control equipment to reduce emissions and comply with environmental regulations. Companies implemented a range of air filtration and abatement technologies—including baghouse dust collectors, electrostatic precipitators, catalytic and thermal oxidizers, and wet and dry scrubbers—to capture and remove airborne contaminants like volatile organic compounds (VOCs), hazardous air pollutants (HAPs), and fine particulates.
The electrostatic precipitator, for instance, represents one of the earliest and most influential pollution control innovations. First conceptualized by German mathematician Dr. M. Hohlfeld in 1824 and later patented by Professor Frederick Gardner Cottrell in 1907, this device uses electrically charged plates to attract and trap airborne particles. Originally intended to recover sulfuric acid mists, electrostatic precipitators have since been widely adopted across power generation, cement production, and chemical processing industries for their efficiency in capturing fine particulates.
The Clean Air Act Amendments of 1990 further expanded regulatory oversight, mandating regular monitoring and reporting of specific criteria pollutants and hazardous emissions from stationary sources. Advanced continuous emissions monitoring systems (CEMS), coupled with accurate sampling equipment and real-time data logging, are now vital for facilities to demonstrate compliance with EPA, OSHA, and other regulatory agencies’ standards. Emissions monitoring not only helps with licensing and environmental audits, but also enables proactive maintenance, operational optimization, and the evaluation of installed air pollution control solutions.
Through the dedication, research, and advocacy of environmental scientists, engineers, and policymakers, significant progress has been made in reducing VOCs, HAPs, and particulate emissions in recent decades. However, as climate change emerges as a global priority, carbon emissions (CO2 and greenhouse gases) have come under increasing scrutiny. Modern regulations now focus on lowering industrial carbon footprints, advancing sustainable air abatement solutions, and promoting energy-efficient pollution control equipment.
In response to evolving standards and the increasing complexity of regulatory requirements, manufacturers may need to go beyond traditional air pollution control systems like incinerators or oxidizers. Advanced solutions such as high-efficiency mist collectors, multi-stage wet and dry scrubbers, modular electrostatic precipitators, regenerative thermal oxidizers (RTOs), and hybrid systems that integrate various filtration and abatement technologies are now at the forefront of industrial air quality management. When selecting pollution control equipment, facility owners and environmental engineers should consider factors like process emissions profile, existing infrastructure, operational costs, local and federal emission limits, as well as the potential for future scalability and integration with energy recovery systems.
Understanding the evolution and application of air pollution control equipment not only supports regulatory compliance but also protects the health of workers, communities, and the environment—creating a cleaner, safer, and more efficient industrial landscape for future generations.
As sustainability and industrial air quality standards gain global momentum, nearly every modern manufacturing or processing facility implements some form of air pollution control equipment to minimize emissions of volatile organic compounds (VOCs), hazardous air pollutants (HAPs), particulate matter (PM), and greenhouse gases (GHGs). These systems are vital for maintaining compliance with environmental regulations, such as those enforced by the EPA and other government agencies through regular inspections, certifications, and monitoring of stack emissions.
Air pollution control equipment manufacturers provide a diverse range of engineered solutions tailored to address the specific air quality challenges found in industries such as chemical production, power generation, pharmaceuticals, automotive, mining, metal fabrication, and food processing. Selecting the optimal air quality control system depends on factors such as emission composition, process temperature, exhaust stream flow rates, and facility layout.
Among the most effective VOCs removal technologies available, carbon adsorbers function by channeling contaminated air over or through a bed of activated carbon. This carbon media traps and retains VOCs, odors, and other airborne toxins, while allowing purified air to pass through. Carbon adsorbers excel at treating emissions containing organic solvents from industries like printing, painting, and chemical manufacturing.
Various adsorbent media can be used to optimize performance for specific applications, including lithium hydroxide, sodium hydroxide, amines (such as monoethanolamine), minerals, and zeolites (e.g., serpentinite). Routine replacement or regeneration of carbon beds ensures continued high-efficiency pollutant removal and cost-effective operation.

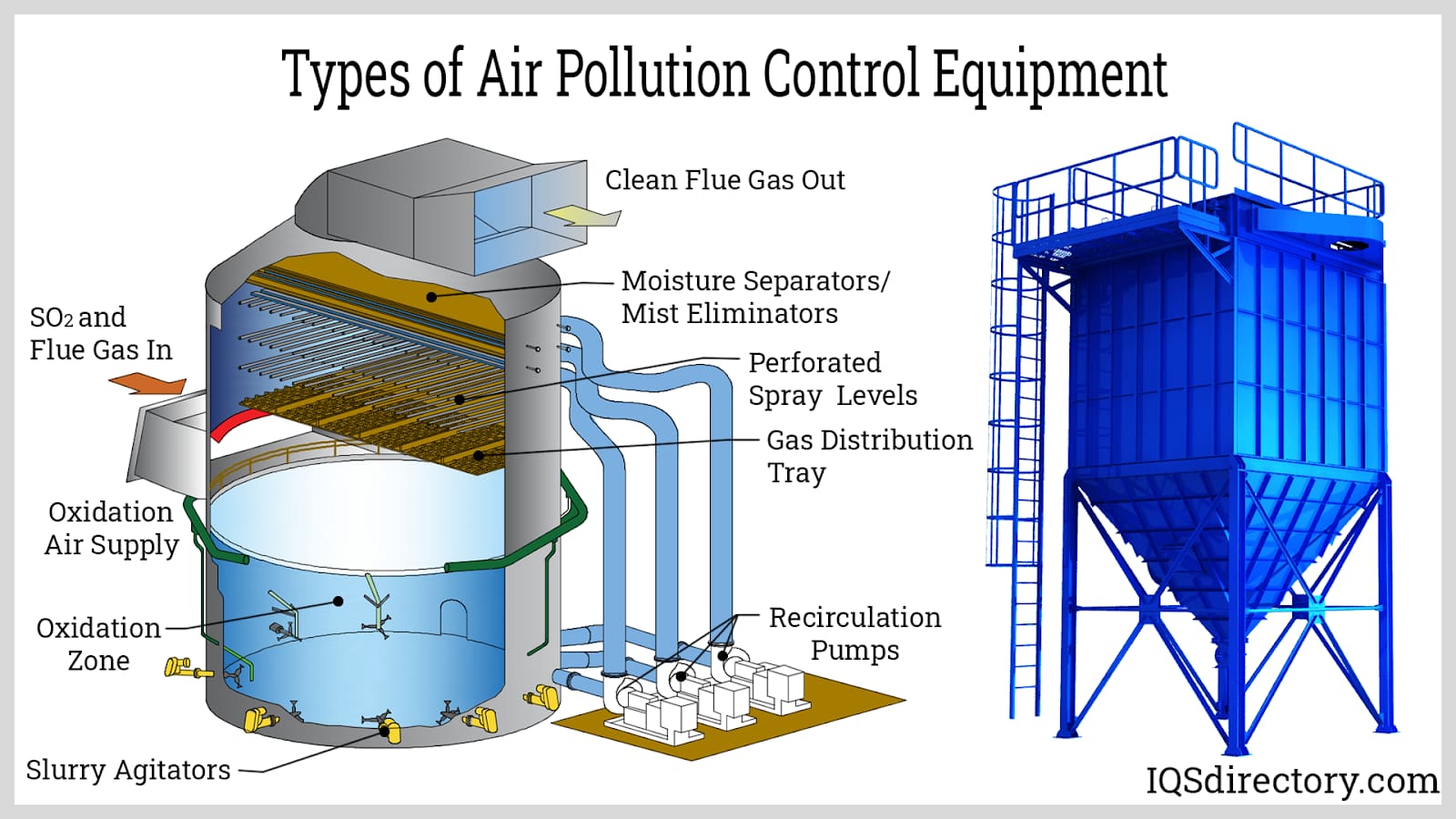
Air scrubbers are advanced air purification systems that capture airborne contaminants, remove particulate matter, and treat toxic or corrosive gases in industrial emissions. By filtering or cooling the airstream as it enters, these systems can effectively reduce harmful particulates, improve indoor air quality, and support compliance with environmental standards. Air scrubbers come in both wet and dry configurations, distinguished by their method of contaminant removal. They are commonly used in chemical processing, petroleum refining, and waste incineration industries where air pollution control is critical.

Wet scrubbers use water or aqueous solvent sprays to capture and neutralize pollutants such as sulfur dioxide (SO2), ammonia, and particulate matter from industrial exhaust streams. Their superior removal efficiency makes them ideal for controlling emissions from applications generating acidic gases, odors, and dust, including wastewater treatment, pulp and paper, and chemical manufacturing facilities. Wet scrubbers often form a critical component of multi-stage pollution control systems designed to address complex emission profiles.
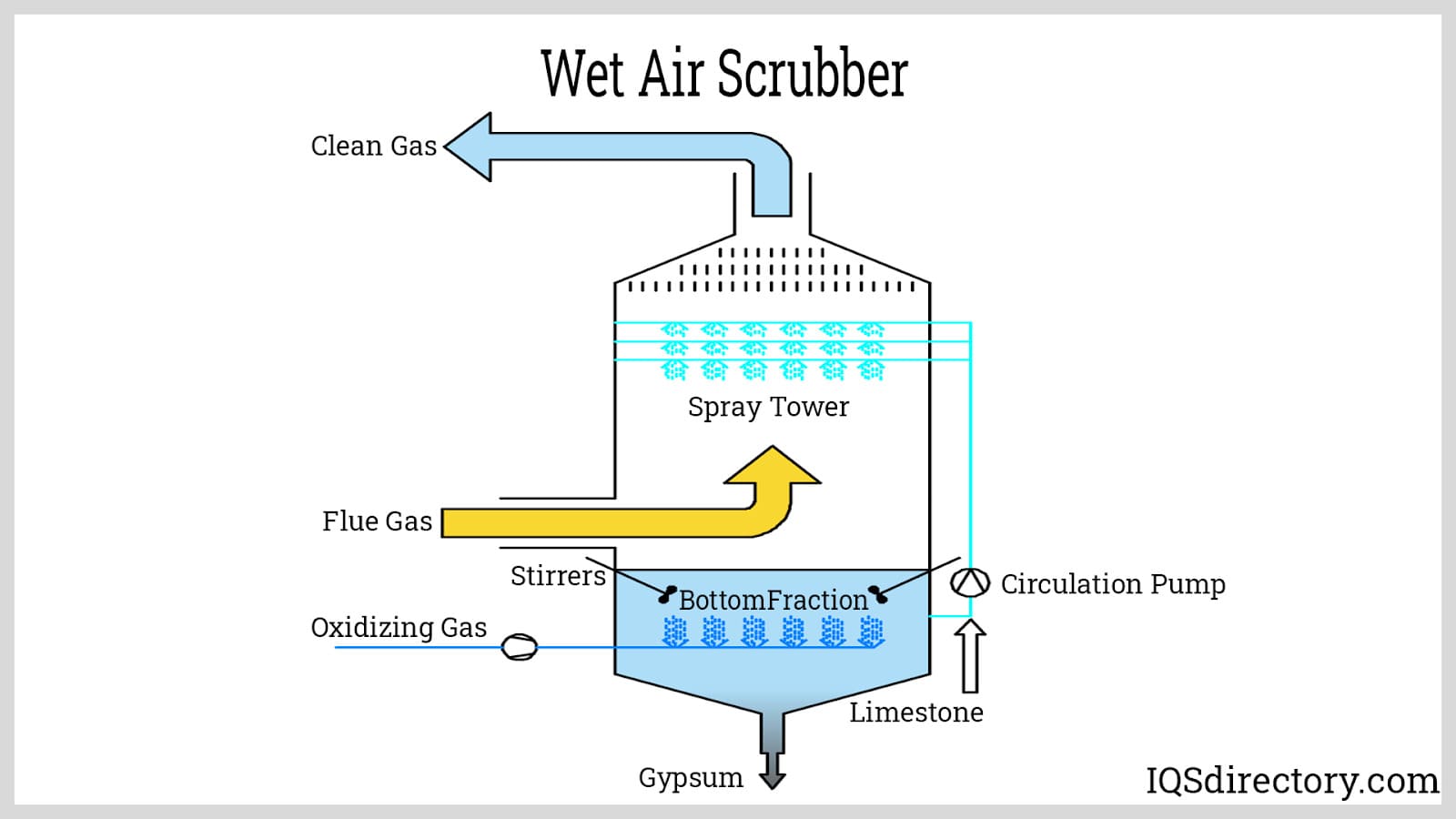
In operation, contaminated air enters the scrubber’s lower chamber, then passes upwards into a packed bed where it is thoroughly mixed with fine solvent mist. A mist eliminator removes residual droplets, ensuring only clean air exits the stack. The solvent’s ability to absorb targeted pollutants depends on factors such as its chemical composition, pH, and ionic charge, which influence bonding with inorganic contaminants.
The adaptability of wet scrubbers allows for removal of multiple pollutant classes—including odors, gases, mists, sulfides, and acidic vapors—making them a preferred air emissions control technology for facilities with fluctuating or mixed pollutant loads.
Dry scrubbers employ chemical reagents—such as lime or sodium bicarbonate—to react with and neutralize gaseous pollutants in industrial exhaust streams. As the process does not require liquid solvents, there is no liquid waste, thereby minimizing disposal and operational costs. Dry scrubbers are frequently integrated into flue gas desulfurization systems and are effective for capturing acidic vapors and soluble hazardous compounds, especially in applications with lower exhaust moisture or volume.
The reaction products—primarily salts and other neutralized byproducts—are captured in a filter system or incinerated by high exhaust temperatures, making dry scrubbers both efficient and eco-friendly for air quality management.

Electrostatic precipitators (ESPs) are sophisticated, filterless devices that use electrically charged plates or wires to extract solid particles—such as dust, soot, ash, smoke, and fine fumes—from industrial gas streams. ESPs offer exceptionally high fine-particle removal efficiencies, making them indispensable for controlling emissions from power plants, manufacturing, steel mills, and any process relying on combustion of fossil fuels.
Contaminated exhaust gases pass through high-voltage electrodes, imparting a static charge to airborne particulates. These charged particles are attracted to collector plates of the opposite polarities, where they accumulate for safe removal and disposal. Electrostatic precipitators are a mainstay of industrial air purification and emission control, reducing health risks, environmental impact, and ensuring compliance with stringent clean air standards.
Furthermore, ESPs can be configured to target a particular size range of particles, adapting to variations in dust loads or process changes. Their modular design also makes them scalable for large facilities or high-throughput operations.

Thermal oxidizers—sometimes referred to as incinerators—destroy dangerous airborne chemicals and organic compounds through controlled high-temperature combustion. These devices transform HAPs and VOCs present in industrial gases into water vapor and carbon dioxide, reducing the release of toxic substances. With advanced safety mechanisms and real-time monitoring, oxidizers are extensively used in large-scale manufacturing, chemical production, and pharmaceutical plants for regulatory compliance and health protection.
Oxidizer system variations (including catalytic oxidizers and regenerative oxidizers) offer trade-offs between energy recovery, operational costs, and compatibility with different pollutants. Modern systems often feature heat recovery solutions—such as air-to-air or ceramic heat exchangers—to maximize fuel efficiency and lower operating expenses while maintaining high destruction rate efficiency (DRE). Wet electrostatic precipitators (WESPs) or downstream scrubbers may be integrated to address the removal of acid gases or particulate matter generated during the oxidation process.
Catalytic oxidizers run at lower operating temperatures than thermal oxidizers, leveraging catalyst-coated media such as platinum or palladium to accelerate the breakdown of VOCs and convert hazardous organic gases into environmentally safe compounds. This process not only minimizes fuel usage but also extends the lifespan of pollution control equipment while achieving regulatory emission limits. These systems are widely adopted in chemical processing, paint and coating, and printing industries where VOC destruction is a priority.
Available in regenerative and recuperative configurations, catalytic oxidizers can recycle heat—reducing the total energy demand of the air emissions control system. Their modularity allows seamless integration into both new and existing industrial pollution control infrastructures across diverse sectors.

Regenerative thermal oxidizers (RTOs) maximize energy recovery by using ceramic heat transfer beds to preheat incoming waste gases via a cyclical process. This technology enables RTOs to achieve DRE rates of over 99% for VOCs, HAPs, and odorous emissions, while dramatically reducing natural gas or fuel input. RTOs are particularly effective in handling high-volume, low-VOC-concentration airstreams typical of processes such as painting, coating, and printing.

Recuperative oxidizers utilize traditional shell-and-tube or plate-type heat exchangers to recover energy released from oxidized gases, pre-heating incoming streams and enhancing overall efficiency. These oxidizers are optimal for applications requiring high thermal efficiency with variable process temperatures, such as chemical reactors, solvent abatement systems, and large-scale manufacturing plants. The integration of heat exchangers facilitates significant reductions in operating costs and heat losses.

Direct-fired thermal oxidizers (DFTOs), also known as afterburners, offer a straightforward configuration for the destruction of hydrocarbon emissions. Without the inclusion of heat recovery systems, these oxidizers achieve rapid pollutant breakdown by maintaining high dwell times and elevated temperatures, making them suitable for processes generating concentrated organic vapors or intermittent emission spikes. With destruction efficiencies reaching up to 99%, DFTOs represent a proven solution for rapid compliance with air emission standards at minimal capital cost.

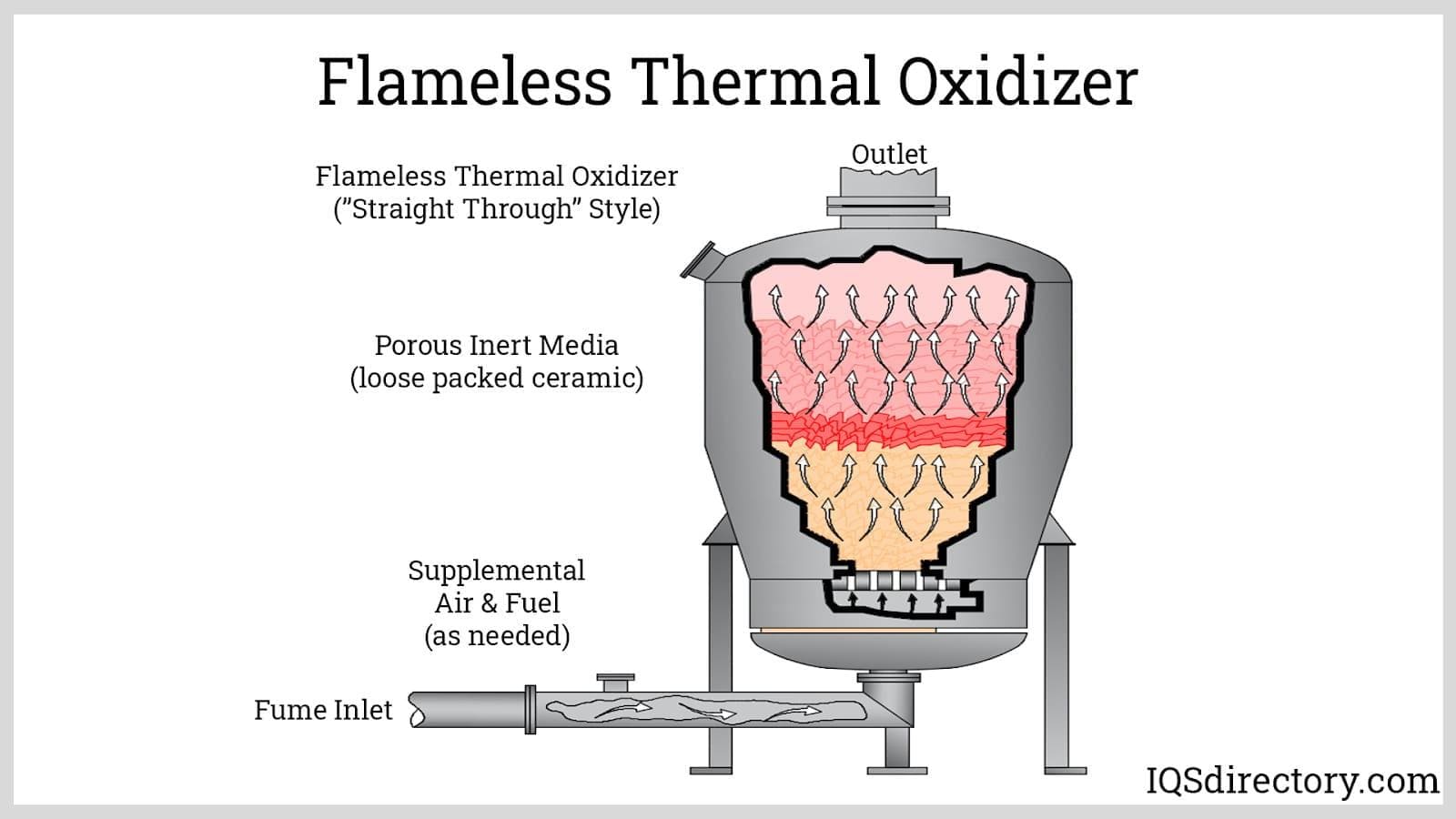
Flameless thermal oxidizers (FTOs) introduce air and waste gases into preheated, non-catalytic ceramic beds to ensure thorough mixing and uniform temperature distribution. By preventing the formation of temperature gradients and hot spots, FTOs optimize the thermal destruction of pollutants like VOCs and maintain ultra-low emissions of nitrogen oxides (NOx). These systems are often adopted in sensitive environments where strict emission limits and process consistency are paramount. When waste heat is insufficient, burners or electric heaters supplement the necessary thermal energy for reliable system operation.
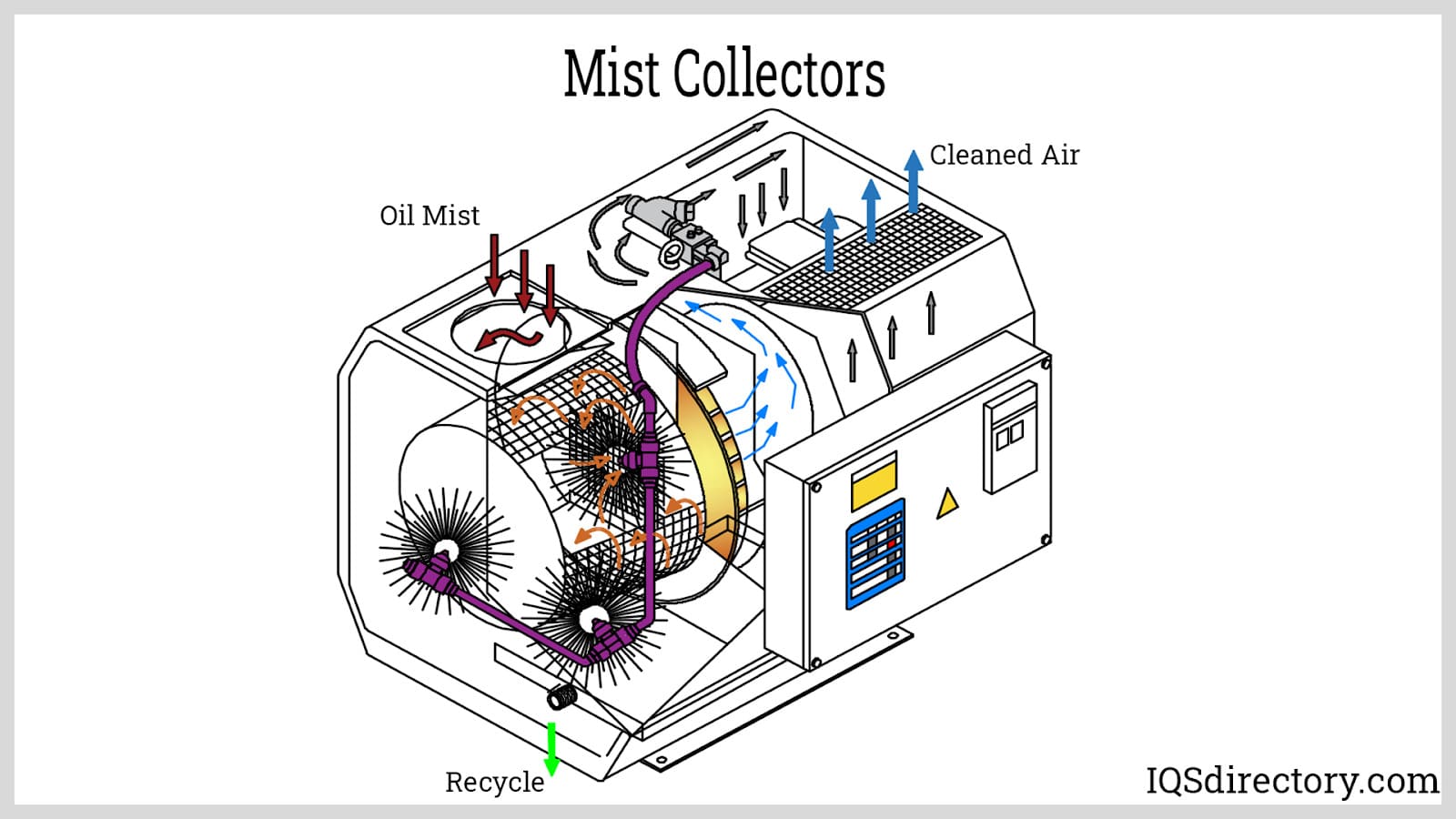
Mist collectors, sometimes called moisture-eliminator filters or demisters, are specially engineered to remove submicron droplets, vapor, and oil mist from exhaust gas streams. Their high filtration efficiency makes them ideal for capturing fine aerosols in metalworking, machining, pharmaceutical, and food processing industries. Mist collectors leverage filter media—often comprised of dense mesh or coalescing pads—to consolidate liquid particles for collection and recovery, often maintaining over 99% efficiency for droplets below 0.3 micron in diameter.
Mist collectors are best suited for non-particulate exhaust applications and environments where gas streams remain below 120 °F (48 °C). For gas streams carrying abrasive particulates or extremely high temperatures, alternative air pollution control technologies are recommended.
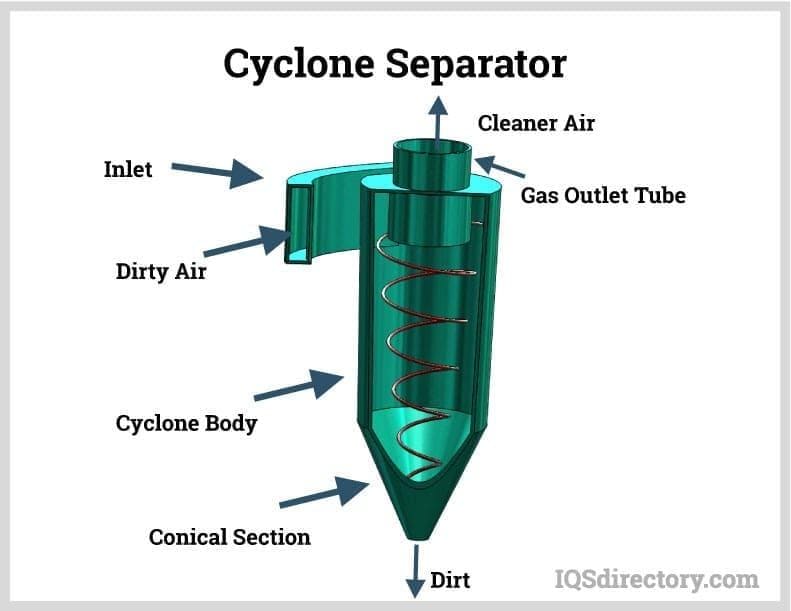
Also referred to as cyclonic separators, cyclone dust collectors efficiently eliminate heavy dust, sawdust, woodchips, and non-fibrous particulates from industrial exhaust without the need for disposable filter media. These systems induce a high-velocity spiral in the gas flow, generating centrifugal force that propels particulate contaminants against the chamber walls for collection. Cyclones are popular as primary dust collectors in woodworking, mineral processing, cement production, biomass energy, and foundry operations. For fine particulate or submicron dust removal, they are often paired with secondary filtration technology, such as baghouses or cartridge collectors.
This modular approach increases air quality system reliability and allows facilities to meet even the most demanding particulate emission limits.
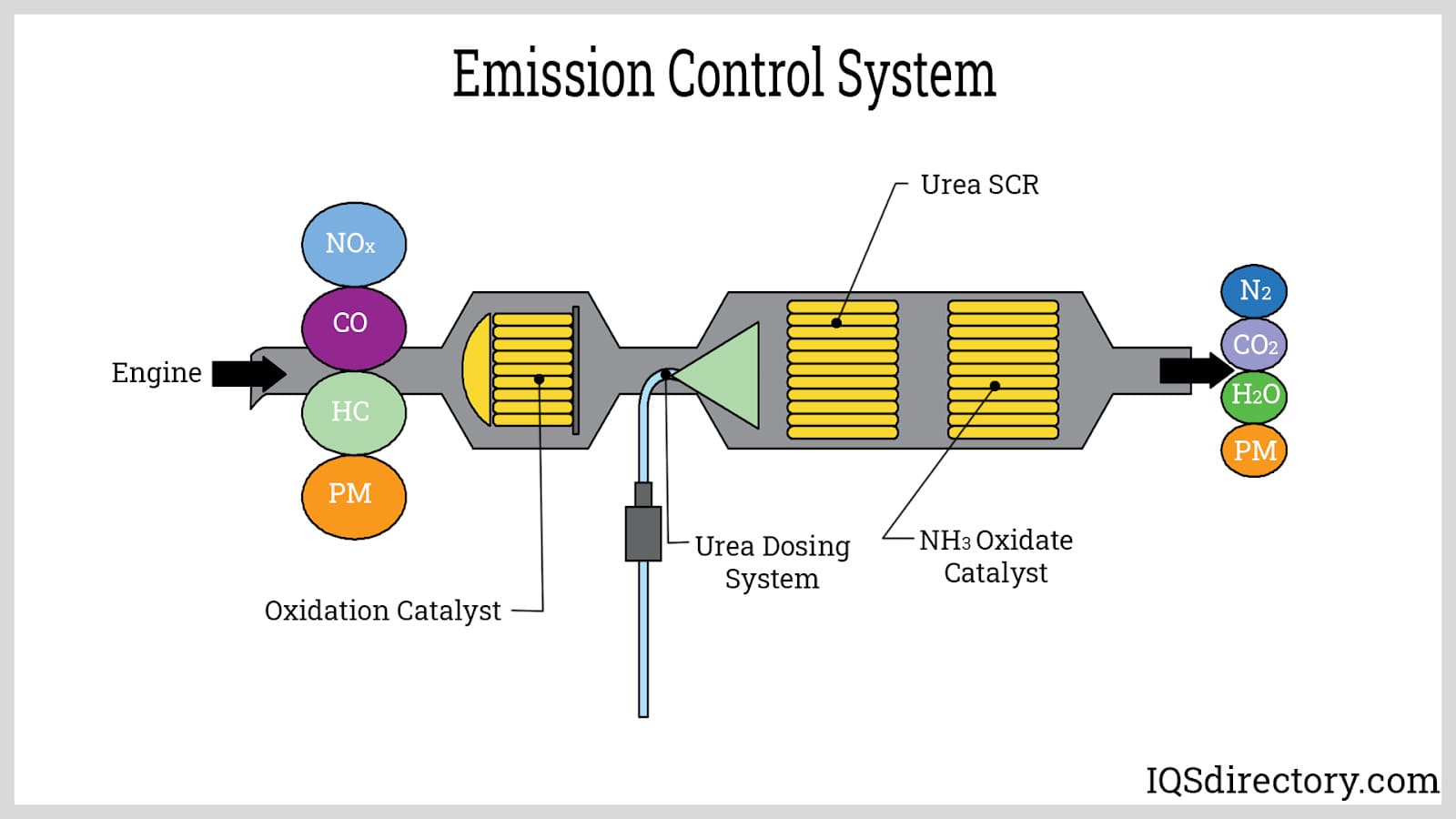
Selective Catalytic Reduction (SCR) systems, or catalytic reactors, represent the industry best practice for reducing high levels of nitrogen oxides (NOx) in fossil fuel combustion exhaust. Industrial SCR units inject ammonia into the flue stream, allowing reactive surfaces with catalysts to convert NOx to harmless nitrogen and water vapor. These reactors may also reduce other gaseous pollutants, including carbon monoxide (CO) and VOCs, as in the case of the three-way catalytic converters used in automotive emission control.
While SCR technology offers over 90% NOx reduction and can attain up to 99.99% efficiency for select gases, it is generally unsuitable for exhaust flows with high dust or particulate content and is comparatively costly compared to filtration-based pollutant control. For integrated air pollution control, SCR units are often combined with particulate abatement stages, such as baghouses or ESPs, in power plants and other stationary combustion sources.
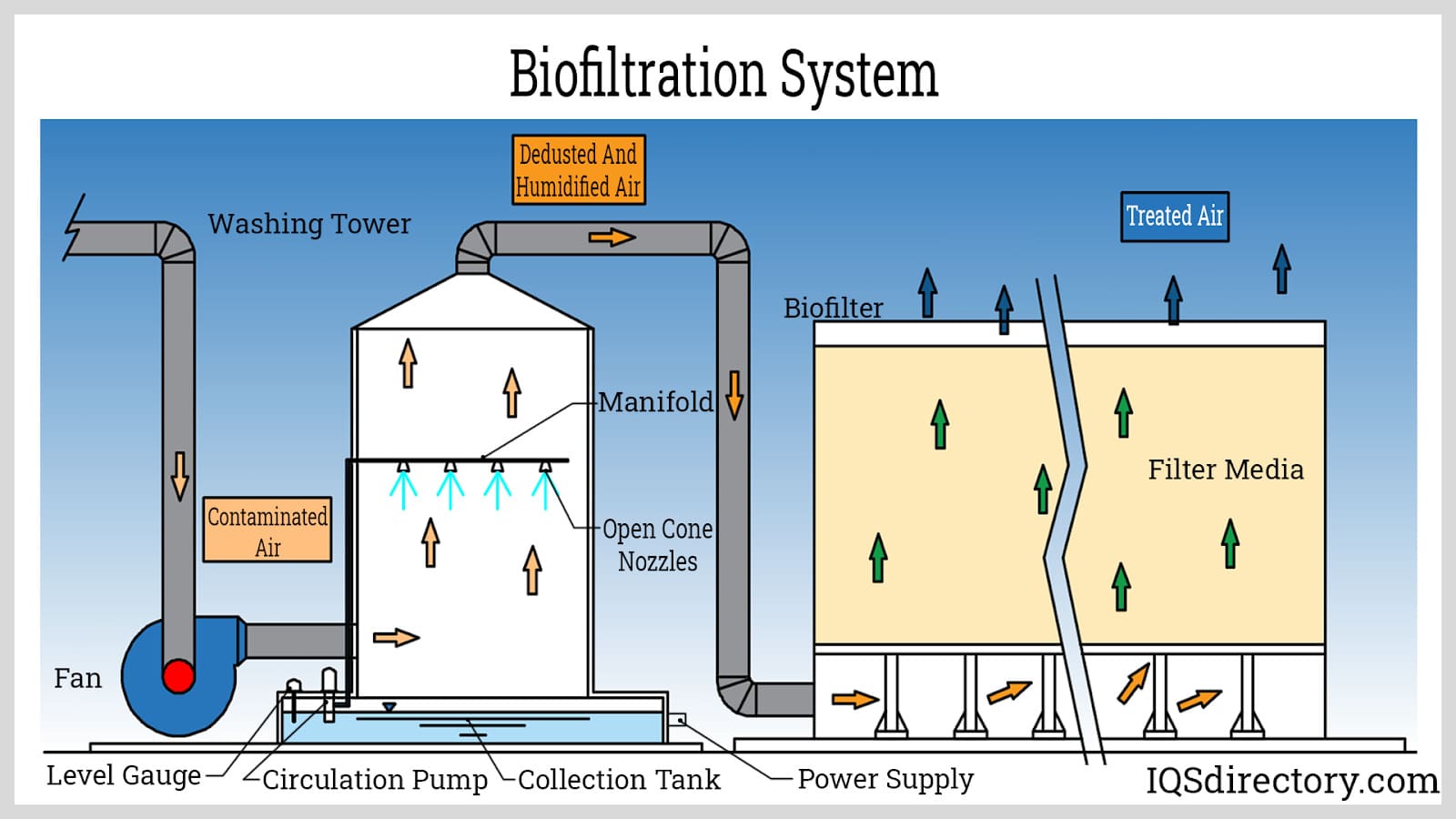
Biofilter systems harness the metabolism of naturally occurring microorganisms—primarily bacteria and fungi—to biologically degrade water-soluble chemicals, organic HAPs, and VOCs from contaminated air. Unlike combustion-based control equipment, biofilters do not generate hazardous byproducts such as NOx or CO. Instead, they convert pollutants into benign end-products, delivering over 98% removal efficiency for various organic contaminants. Typical installations include compost-based beds or engineered synthetic media, and biofilters are optimal for industries such as municipal wastewater plants, food processing, composting, and certain chemical production operations.
By understanding the performance characteristics and application requirements of each air pollution control equipment type, companies can engineer integrated air quality management systems that address evolving regulatory, operational, and environmental challenges—protecting employees, communities, and the global climate. When selecting the best emission control solution, factors such as contaminant type (organic vs. inorganic), particulates vs. gases, process flow rates, operational temperature, regulatory limits, and cost of operation must all be carefully considered. Consulting with experienced air quality engineers and evaluating third-party equipment reviews can help ensure compliance and optimal performance of your facility’s pollution control system.
Air pollution control equipment consists of systems designed to limit the release of solid and gaseous pollutants into the atmosphere from industrial sources, primarily focusing on managing acidic gas emissions and particulate matter.
Such equipment operates through methods like chemical alteration (neutralizing hazardous gases), hazard elimination (using air filters), and advanced processes such as catalytic and thermal oxidation, filtration, and biofiltration to efficiently remove contaminants.
Key equipment types include carbon adsorbers, air scrubbers, wet and dry scrubbers, electrostatic precipitators, oxidizers (thermal, catalytic, regenerative, recuperative, flameless), mist collectors, cyclone dust collectors, catalytic reactors, and biofilters.
Continuous monitoring ensures compliance with environmental regulations and provides operational data for proactive maintenance, system optimization, and accurate reporting of hazardous emissions to regulatory agencies.
Selection criteria include the physical and chemical properties of pollutants, exhaust composition, monitoring compatibility, regulatory requirements, system maintenance, process temperature, flow rates, and facility layout.
Major incidents in London and Donora, Pennsylvania highlighted the dangers of unchecked emissions, leading to landmark legislation such as the UK’s Clean Air Act (1956) and the US Clean Air Act (1970), thus driving the adoption of advanced control technologies globally.
Industrial air pollution control equipment is crucial and must be prioritized. Almost every industry contributes to environmental pollution through its operations. Sectors such as petroleum, oil, coal, metal, chemical, and waste management are among the major contributors to toxic emissions.
Industrial activities—including raw material extraction, product manufacturing, site and machinery maintenance, and transportation—inevitably generate pollution. Burning fossil fuels releases volatile hydrocarbons, while the use of wood and coal as fuel results in carbon dioxide and sulfur dioxide emissions. Additionally, automobiles contribute significantly to harmful carbon emissions. Each industrial process emits pollutants that contaminate the air, soil, or water.
Non-industrial air pollution control technologies are also used in homes, cars, and other mobile environments. For example, home air conditioners often feature filtration systems that remove impurities such as pet dander, allergens, mold spores, and dust.
Additionally, precision filtration systems help reduce vehicle emissions from engines, exhaust systems, and air conditioning units.
Many facilities use continuous emissions monitoring systems (CEMS) to track, manage, and report emissions. These systems utilize various instruments to measure the concentration of particulate matter and gaseous chemicals at specific points, often in stacks or ducts. They also assess physical properties of waste gas streams, such as opacity. The New Source Performance Standard (NSPS) and the New Source Review (NSR) require emissions monitoring at major pollution sources, and certain EPA regulations also mandate continuous emissions monitoring.
In addition to parametric monitoring, continuous emissions monitoring helps technicians comply with Compliance Assurance Monitoring (CAM) regulations.
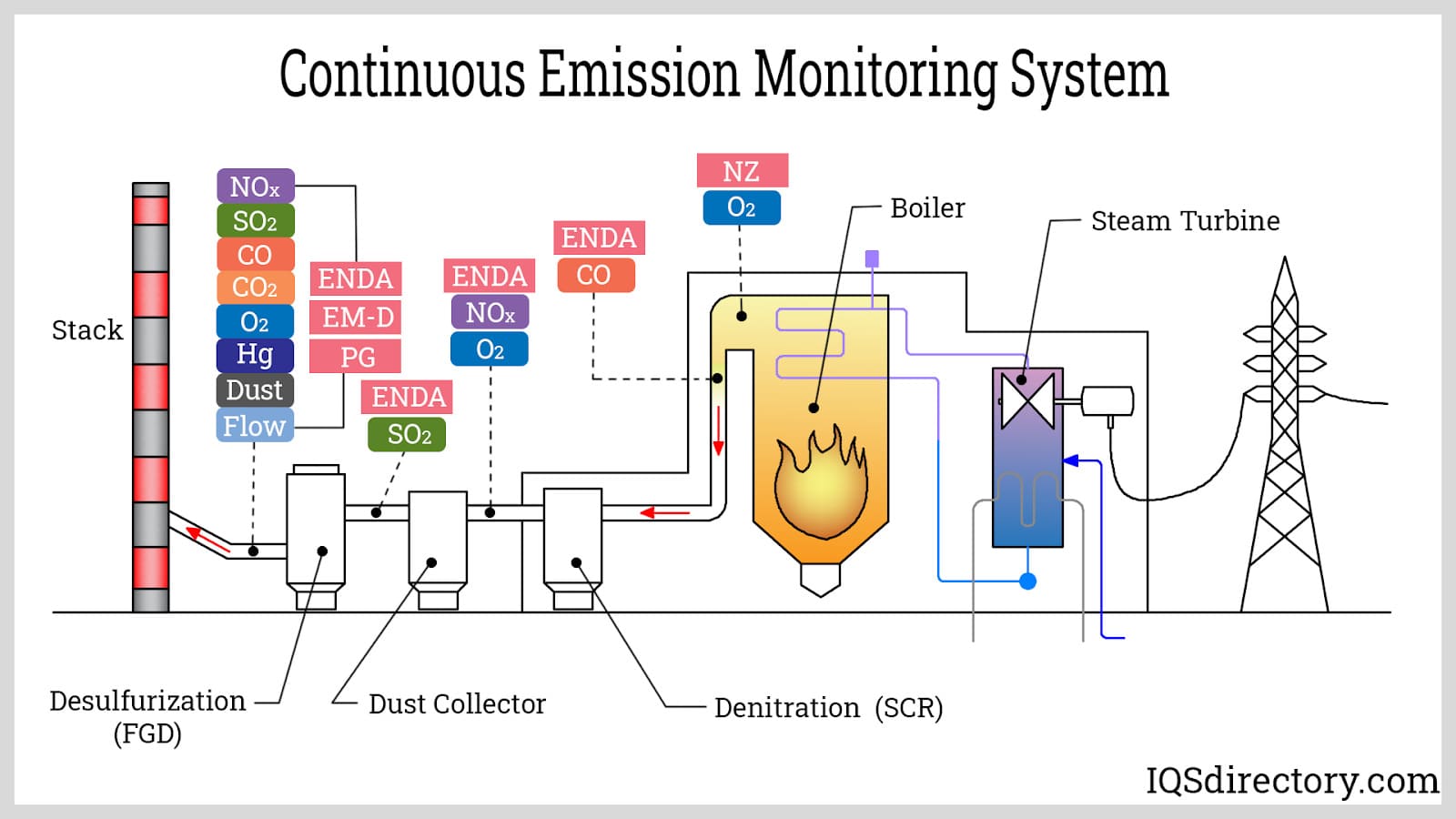
Parametric monitoring measures emissions by tracking key parameters related to the operation of process or air pollution control equipment. This method uses pollutant emission levels and monitored control parameters to assess compliance. The adoption of CAM regulations has increased the acceptance of parametric monitoring, as it offers a more flexible and cost-effective approach to demonstrating compliance.

An air scrubber is a purification system designed to remove particulate matter from the air. It achieves this by using moisture or by cooling and filtering the airstream as it passes through the scrubber.

Air pollution refers to any unwanted particles in the air that contaminate the environment and harm the health of living organisms. These particles can be toxic and may lead to a range of serious health issues, including...
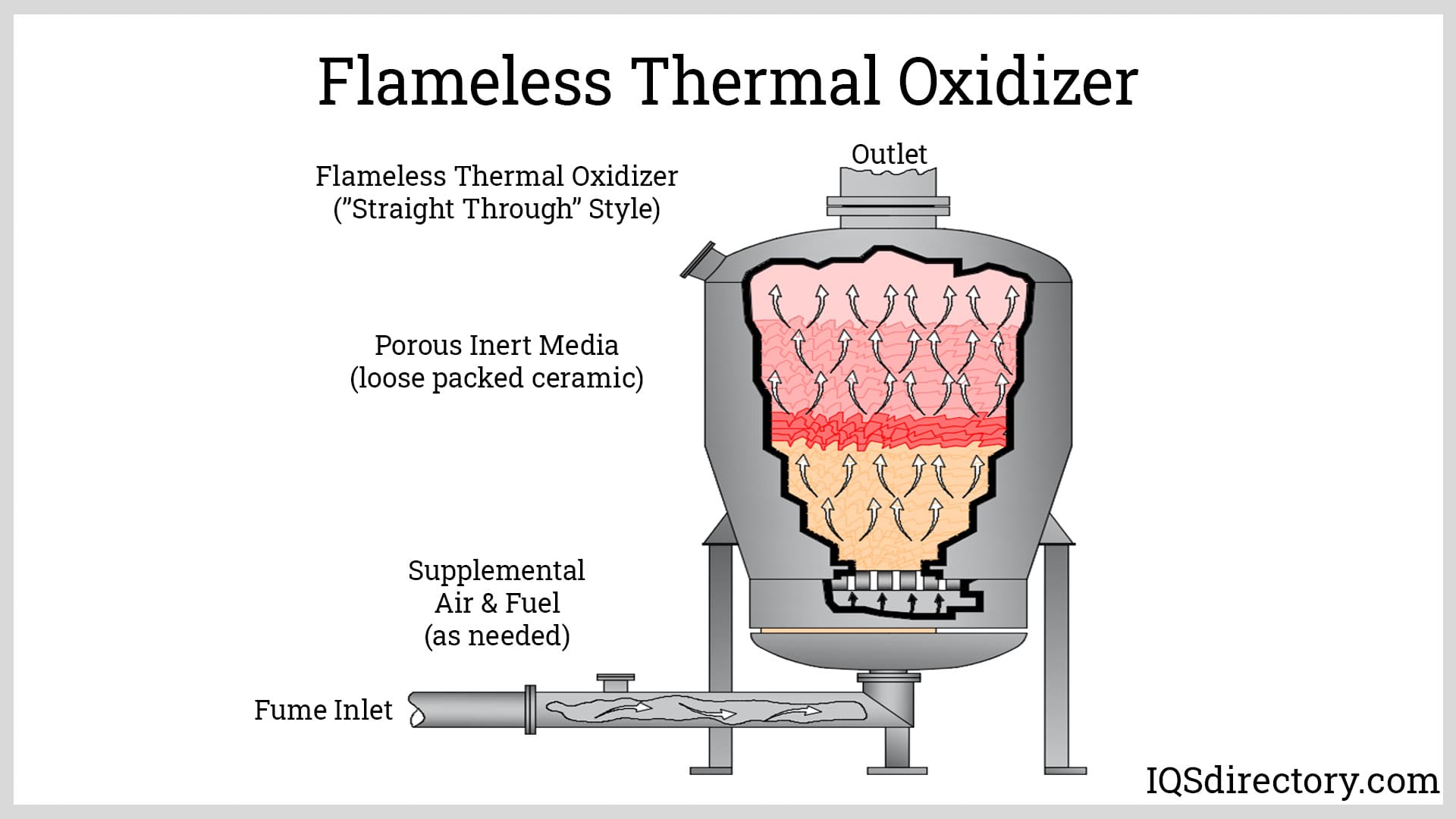
Oxidizers, also known as incinerators, are devices used to treat waste gases or emissions from industrial processes by thermally breaking down harmful pollutants into simpler, more stable compounds.
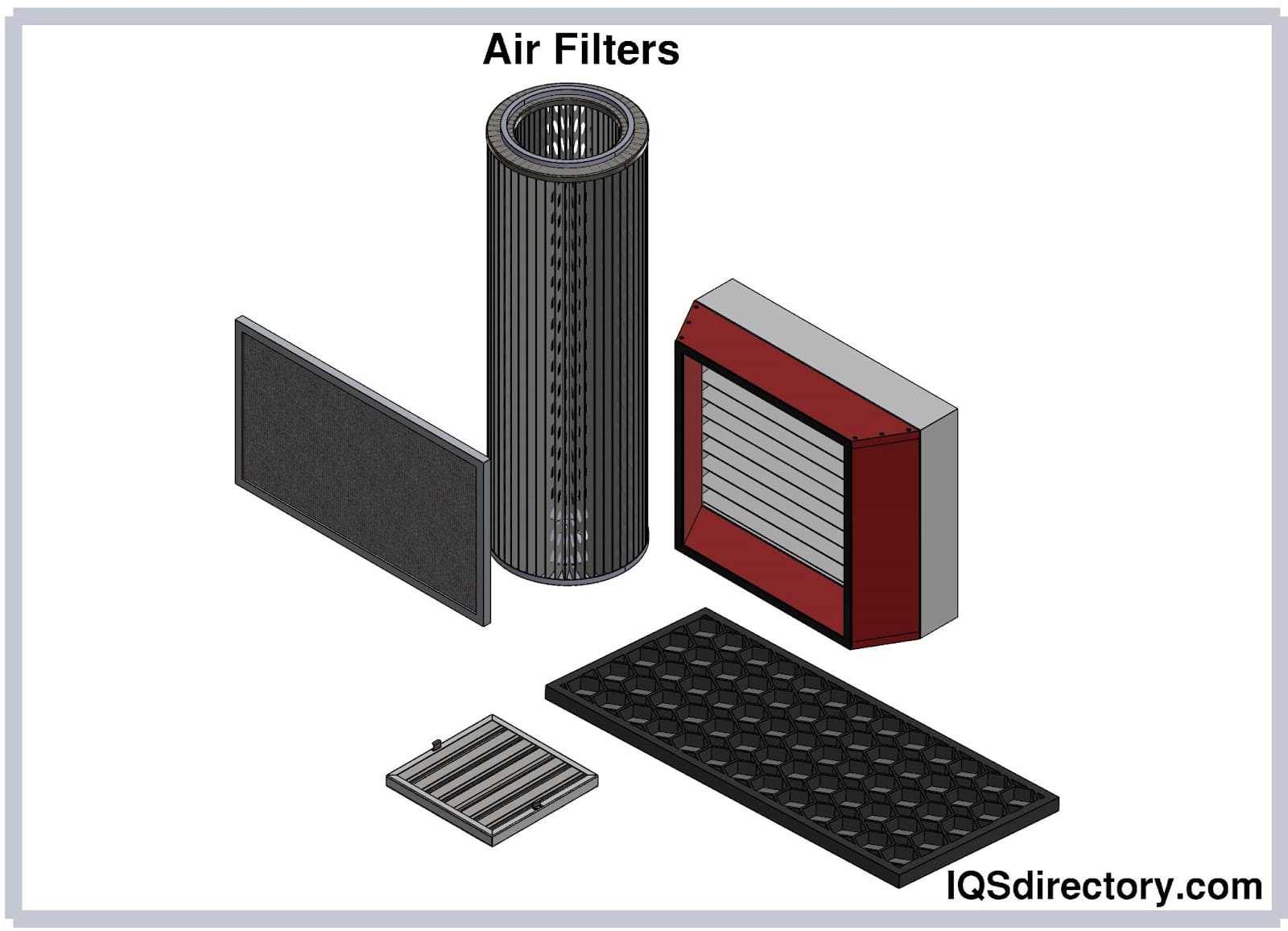
Air filters are devices used to remove airborne particles, pollutants, and microorganisms hazardous to health and the ecosystem. In industrial facilities, air filters preserve the quality of products and materials and protect critical equipment from damage...

A baghouse is a pollution control system designed to remove, capture, and separate dust, particulate matter, and dirt from the air in manufacturing or processing facilities. It utilizes filters in the form of tubes, envelopes, or cartridges. The primary components of a baghouse are the filter media or bags, which trap particles as air flows through the system.
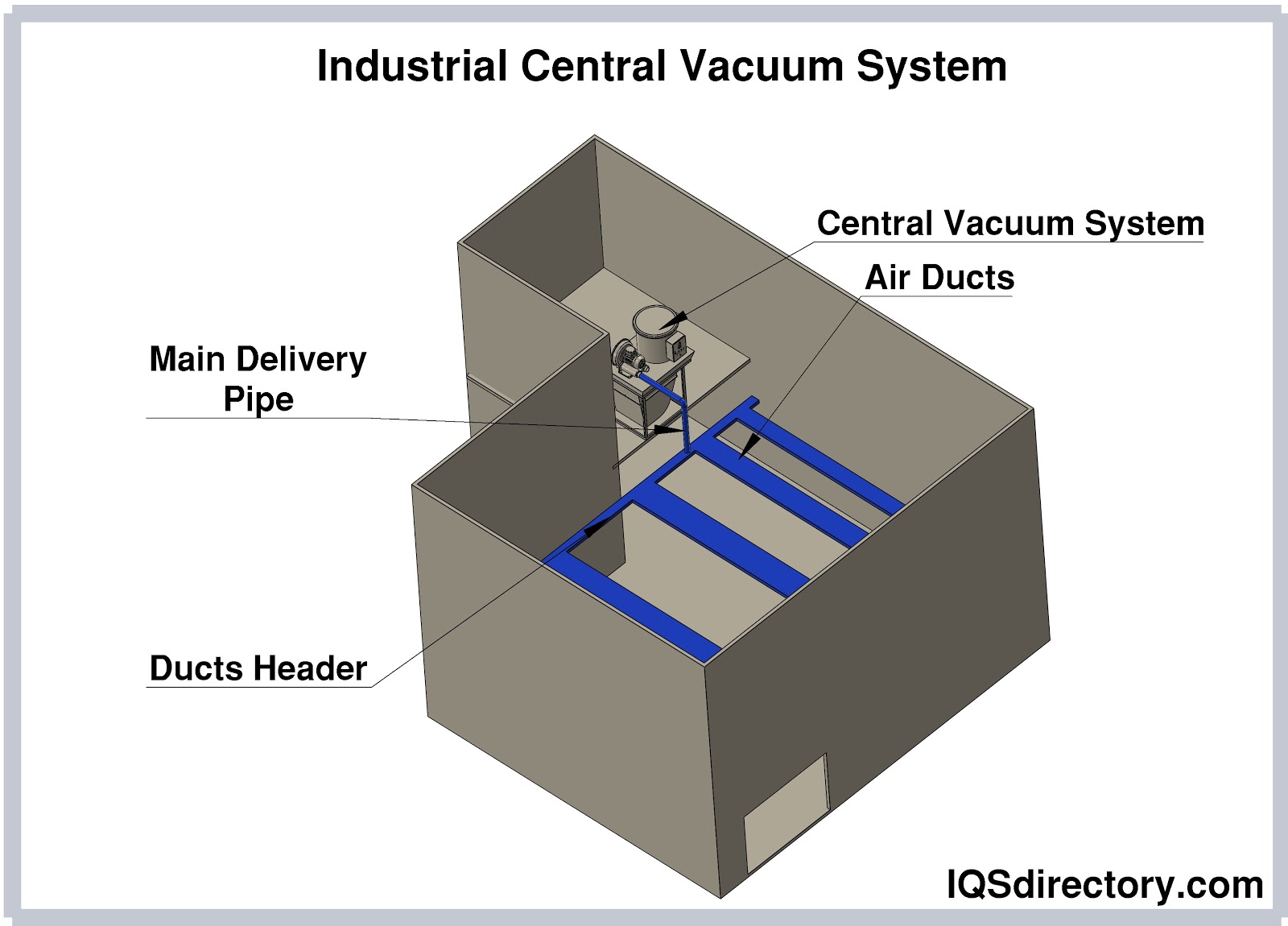
A central vacuum system is a cleaning mechanism that is built into a building for ease of use, access, and maintenance. Vacuum and pressure are created by a centrally located motor to remove dirt, dust, and debris...

A dust collection system is designed to remove particulate contaminants from the air in production facilities, workshops, and industrial complexes. It cleans the air by directing it through a series of airtight filters.

Explosion-proof vacuums are designed to safely handle potentially flammable environments. They use compressed air, electricity, and cyclonic motion to prevent the ignition of gases or vapors. These vacuums operate at temperatures that avoid igniting a potentially explosive atmosphere, and their motors are replaced with specialized, safety-certified components to ensure safe operation.

A HEPA filter is a high-efficiency, pleated air filter designed to capture very small particles, down to the size of a micron (µ), which is 1/1,000th of a meter.
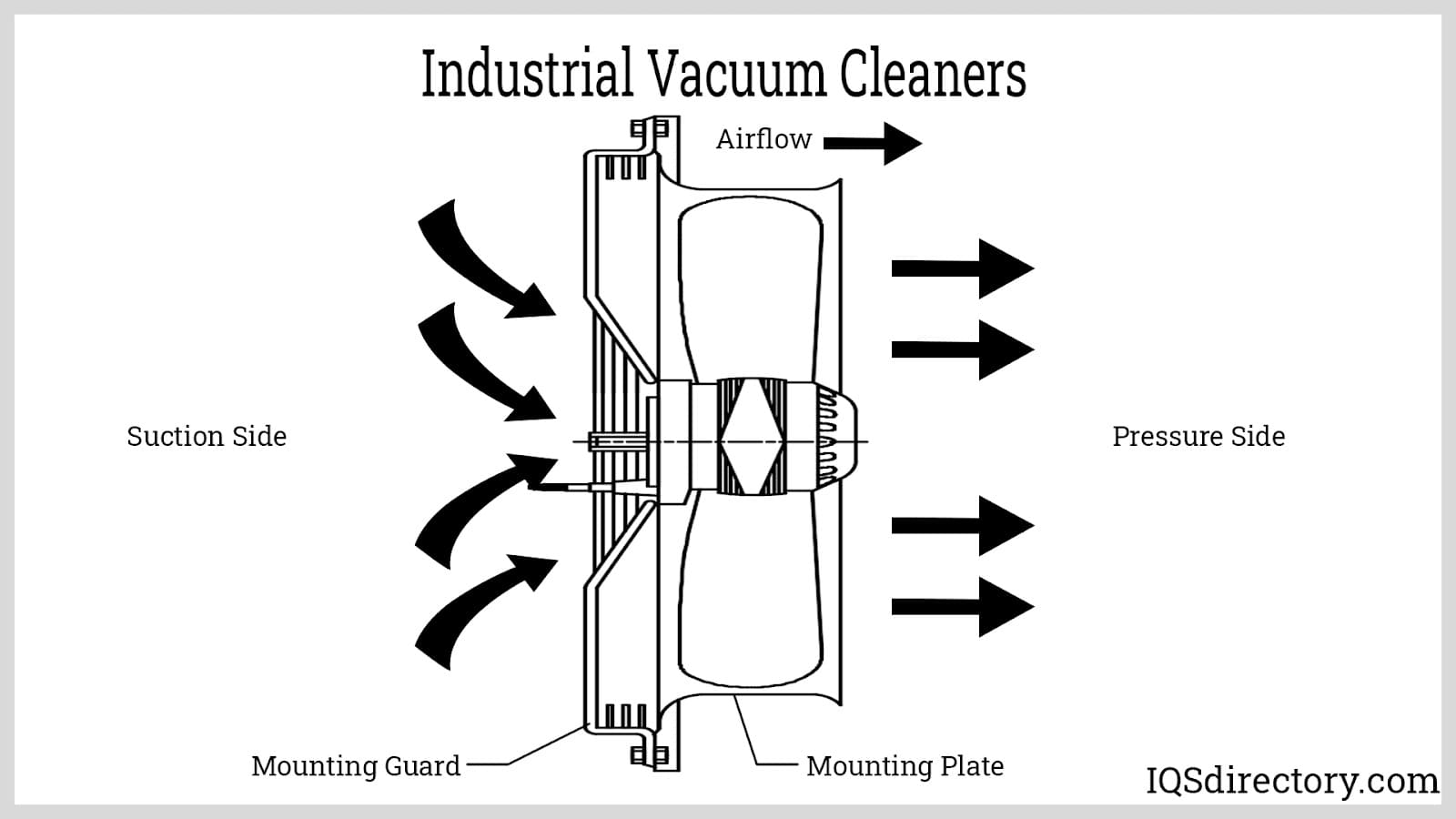
An industrial vacuum cleaner is a robust cleaning device engineered to handle debris, industrial waste, construction refuse, and residual materials left over from manufacturing or construction activities.

A jet dust collector is a suction filtration unit that draws dust-laden air into a filtration system, capturing particulate matter and expelling clean air. The filters in jet dust collectors are cleaned using compressed air, which helps maintain their effectiveness.

During the Industrial Revolution, as industries started generating large amounts of industrial waste, including fine dust and wood particles, S.T. Jones from America applied for the first dust collector patents in 1852. His invention was a single-bag filter designed to address these issues.
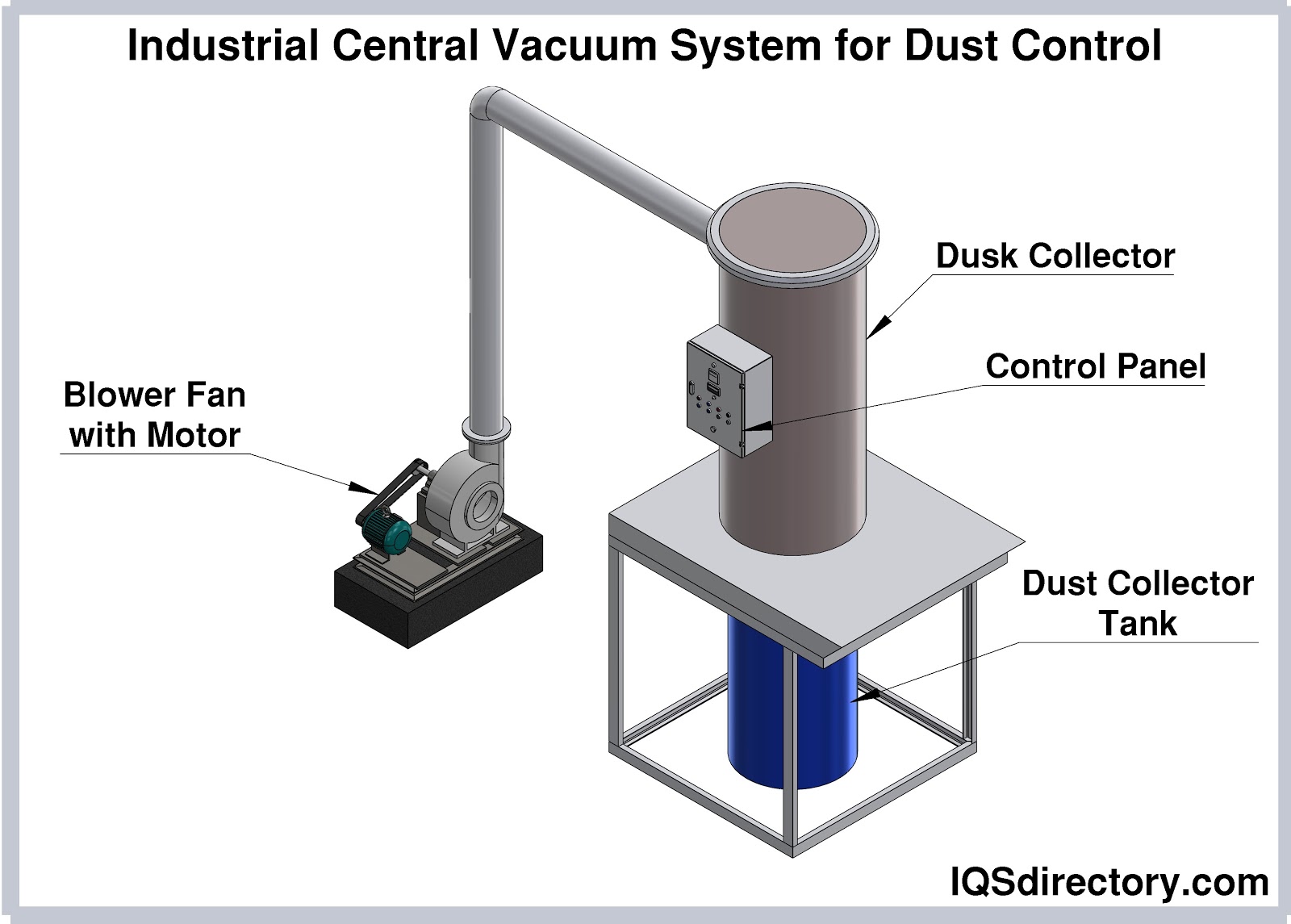
Vacuum cleaners employ suction to gather dirt, dust, waste products from industrial processes, and other debris for disposal, recycling, or reuse. They are primarily used for maintaining and cleaning industrial spaces.