Aluminum Anodizing
Anodizing is an electrolytic passivation process used to increase the thickness of a metal’s natural oxide layer. In aluminum anodizing, that controlled electrochemical reaction strengthens the protective surface that forms on aluminum alloys, creating a finish valued for corrosion resistance, appearance, and long-term durability in manufacturing, architectural, and industrial applications.
During the anodizing process, the workpiece becomes the anode in an electrical circuit. The resulting oxide layer improves paint and adhesive bonding, boosts wear resistance, and helps protect the substrate in demanding environments. While anodizing can also be used on titanium, magnesium, zinc, niobium, hafnium, zirconium, and tantalum, aluminum remains the most common choice because it responds well to the process and gains a stable, tightly bonded oxide finish that helps limit damage caused by alloying elements such as copper and iron.
Aluminum Anodizing FAQs
What is aluminum anodizing?
Aluminum anodizing is an electrolytic passivation process that thickens the metal’s natural oxide layer. It improves corrosion resistance, surface hardness, paint and adhesive bonding, and finish durability while preserving aluminum’s lightweight performance and overall dimensional stability.
What are the main types of aluminum anodizing?
The three primary categories are Type I chromic acid anodizing, Type II sulfuric acid anodizing, and Type III hard anodizing. Type I is often selected for lighter coatings, Type II is common for general-purpose finishing and coloring, and Type III is chosen when thicker, harder, more wear-resistant coatings are needed.
How does clear anodizing differ from color anodizing?
Clear anodizing preserves the natural metallic appearance of aluminum while improving corrosion and abrasion resistance. Color anodizing adds dyes or metallic salts before sealing, allowing decorative or brand-specific finishes while still delivering the protective advantages of the anodized oxide layer.
What materials besides aluminum can be anodized?
Although aluminum is the most frequently anodized metal, titanium, magnesium, zinc, niobium, hafnium, zirconium, and tantalum can also be anodized for added corrosion resistance, color development, surface protection, or decorative performance in specialized applications.
Where is anodized aluminum commonly used?
Anodized aluminum is widely used in electronics housings, cookware, architectural components, automotive trim, aerospace parts, and industrial equipment. Buyers often choose it when they need a lightweight metal finish with strong corrosion resistance, attractive appearance, and dependable wear performance.
Why is sulfuric acid used in anodizing?
Sulfuric acid is used in Type II and Type III anodizing because it helps produce dense, uniform oxide layers that perform well in clear, dyed, and hard-coat applications. It supports consistent finishing, good corrosion protection, and reliable wear resistance across a broad range of aluminum parts.
What determines the color of anodized titanium?
The color of anodized titanium is determined by oxide thickness, which changes with voltage during processing. As light reflects through that oxide layer, it creates vivid interference colors without the need for traditional dyes, making titanium anodizing popular for jewelry, dental, and decorative uses.
History of Aluminum Anodizing
In 1923, oxalic acid anodizing based on an earlier chromic-acid method was first used to help protect seaplane components and architectural materials against corrosion. The process was initially patented in Japan and later adopted in Germany. In 1927, Gower and O’Brien introduced the first sulfuric acid anodizing process, helping shape the aluminum finishing methods that are still widely referenced today.
During the 1960s and 1970s, anodized aluminum gained broad use in architectural applications because of its clean appearance, low weight, and weather resistance. Over time, it faced competition from less expensive materials such as plastics and powder coatings, while phosphoric acid anodizing emerged as an important pretreatment option for adhesives and organic paint systems. Today, aluminum anodizing remains a widely used finishing process for parts that need appearance, protection, or both.
- Modern anodizing standards focus on coating quality, finish consistency, oxide thickness, sealing performance, and end-use durability, rather than on one chemistry alone.
Applications for Anodizing Aluminum
Anodizing does not increase the base strength of aluminum, but it does improve corrosion resistance, surface durability, lubricity, and adhesion. That combination makes anodized aluminum a practical choice across industries where buyers compare appearance, outdoor performance, maintenance requirements, and coating life before selecting a finish. Common applications include:
- Electronics: Used for durable outer housings, panels, heat-dissipating enclosures, handheld devices, camera bodies, and satellite components where low weight and clean finish quality matter.
- Food and Beverage: Common in cookware such as pans, pots, mixers, and processing components because anodized aluminum offers a tough, easy-to-maintain surface.
- Architecture: Applied to window frames, doors, curtain wall components, roofs, railings, trim, and exterior panels that need long-lasting appearance and weather resistance.
- Automotive: Selected for trim, exposed housings, accessory parts, performance components, and decorative surfaces where abrasion resistance and visual consistency are both important.
- Industrial Manufacturing: Used on conveyors, scales, machine components, electrolytic capacitors, protective enclosures, and custom-fabricated parts that benefit from a harder, more corrosion-resistant finish.
Anodized Aluminum Process
Anodized aluminum surfaces are harder than untreated aluminum and can offer low to moderate wear resistance depending on oxide thickness, alloy selection, and sealing method. The anodic film is tightly bonded and more integrated with the base metal than many paints or metal platings, although it can also be more brittle under certain conditions.
When engineers or purchasing teams compare anodized finishes, they often evaluate coating thickness, corrosion resistance, color options, electrical properties, dimensional change, and expected service environment. The most common anodizing categories include:
- Clear Anodize – Type II
- Color Anodize (including black anodizing) – Types I, II, and III
- Titanium Anodize: Similar in concept to aluminum anodizing, titanium anodizing is known for producing a broad color range without traditional dyes.
Type I Anodizing Process
Type I chromic acid anodizing is one of the oldest anodizing methods and is used where lighter coatings and specific performance characteristics are desired. In this electrolytic process, the metal with an attached anode is immersed in a chromic acid solution. As direct current passes through the bath, oxygen forms at the anode and builds the oxide film, while hydrogen is produced at the cathode. Type I can reduce aluminum thickness by roughly 0.02 to 0.4 mils, and it differs from Types II and III in that the direct-current voltage is typically raised gradually during processing. It is also subject to tighter environmental controls because chromic acid emissions require careful handling.
Type II Anodizing Process
Type II anodizing uses sulfuric acid and is widely selected for general-purpose aluminum finishing. It supports clear anodizing, dyed finishes, decorative applications, and many commercial or industrial parts that need an effective balance of corrosion protection, appearance, and coating economy.
Type III Anodizing Process
Type III is also sulfuric acid based but is commonly called hard anodizing or hard coat anodizing. It is performed at lower temperatures and higher current density than Type II, promoting greater anodic growth and producing a thicker, harder, more wear-resistant surface for demanding service conditions.
Custom anodizing processes can also be developed to meet part-specific requirements, such as controlled conductivity, color uniformity, dimensional tolerance, or application-driven hardness and sealing needs.
Clear Anodizing
-
Uses for Clear Anodizing
Clear anodizing can also serve as the foundation for selected dyeing and decorative effects, but it is especially valued when buyers want the natural metallic look of aluminum to remain visible. Typical uses include:
- Architecture: Window and door frames, railings, storefront systems, trim, siding, and other exposed building products.
- Automotive: Trim, exposed housings, interior details, and exterior accessory parts.
- Printing: Commercial photolithography plates and related aluminum components.
- Industrial Manufacturing: Sheet metal, extrusions, profiles, machine guards, and protective cases.
- Jewelry and Artwork: Decorative, display, and functional aluminum pieces that benefit from a clean metallic finish.
The sulfuric acid anodizing process used for clear anodizing offers good wear resistance compared to chromic acid anodizing, along with strong corrosion performance and finish consistency. Other benefits include lower maintenance, light metal weight, attractive appearance, and efficient production for many aluminum parts and extrusions.
-
Clear Anodizing Process
Clear anodizing follows the same general sequence as Type II sulfuric acid anodizing. The metal is immersed in sulfuric acid, which acts as the electrolyte, and the surface is converted into a hard aluminum oxide layer as direct current passes through the bath.
During processing, the oxide film grows from the aluminum surface and develops the porous structure that later allows sealing or coloring when required. Once anodizing is complete, the part is removed from the bath and commonly sealed with hot water or another suitable seal to improve corrosion resistance and stabilize the finish.
If a colored finish is desired, dyes or salts can be introduced after anodizing and before sealing so the color becomes locked into the porous oxide layer. Clear anodizing, however, is often selected specifically to preserve the natural luster of the substrate. While aluminum is the most common metal in this category, titanium, magnesium, zinc, niobium, and tantalum can also be anodized for related surface-performance goals.
Color Anodizing
-
Color Anodizing Dye Range
Color anodizing is a popular way to create long-lasting decorative and functional finishes on metal products. Instead of sitting loosely on the outside like a paint film, the color is introduced into the porous oxide layer before sealing, which helps it resist scratching, peeling, and normal wear. Aluminum and titanium are among the most common metals used for this process because of their versatility and finishing range.
The anodized surface is dyed before final sealing, making color anodizing especially useful where visual appearance, product identification, or brand differentiation matters. A broad range of shades can be produced, from light decorative tones to darker industrial finishes, although some alloy chemistries can make lighter colors harder to achieve consistently.
Color anodizing is widely used because it allows manufacturers to offer many finish options while still keeping the corrosion resistance and wear advantages of anodizing. Buyers comparing black anodized aluminum, bronze anodized aluminum, or custom dyed finishes often look at fade resistance, outdoor exposure, gloss level, and alloy compatibility.
Chemical systems such as ferric ammonium oxalate on anodized aluminum can produce black and gold tones. Inorganic color systems are often more lightfast than many organic dyes, while metallic hues, though more limited in shade range, typically hold their brightness longer and are often used in architectural applications.
The Process for Color Anodizing
The dyeing sequence varies based on the metal, the anodizing type, the desired finish, and the dye chemistry being used. Some metals, including titanium, can develop color through oxide thickness alone without additional dyes, with the final hue changing according to electrical current and film thickness.
After color is introduced, the surface is sealed with hot water or steam, often with nickel acetate, converting the oxide into a more hydrated form. This step helps reduce color bleeding, improves corrosion resistance, and locks the finish into the coating. Whether used for consumer products, industrial parts, architectural metal, or custom components, color anodizing remains popular because it combines durability with strong visual appeal.
Titanium Anodizing
Titanium is frequently anodized for jewelry, decorative items, dental implants, body jewelry, wedding bands, art objects, and specialty consumer products. As the titanium surface changes during anodizing, the oxide layer develops in a way that supports vivid color effects and strong finish stability. In applications where appearance and biocompatibility matter, titanium anodizing is often considered alongside aluminum finishing and decorative coating options.
The resulting color depends on oxide thickness. Light reflecting from both the oxide layer and the underlying metal creates interference colors that can appear bright, varied, and durable without relying on a traditional dye system.
Magnesium Anodizing
Magnesium anodizing is commonly used as a paint primer or pretreatment and can be sealed with materials such as oil or wax. Although zinc is anodized less often, specialized processes have been developed to create hard, corrosion-resistant anodized zinc finishes, including olive-green surface treatments for selected industrial needs.
Aluminum Anodizing Images, Diagrams and Visual Concepts
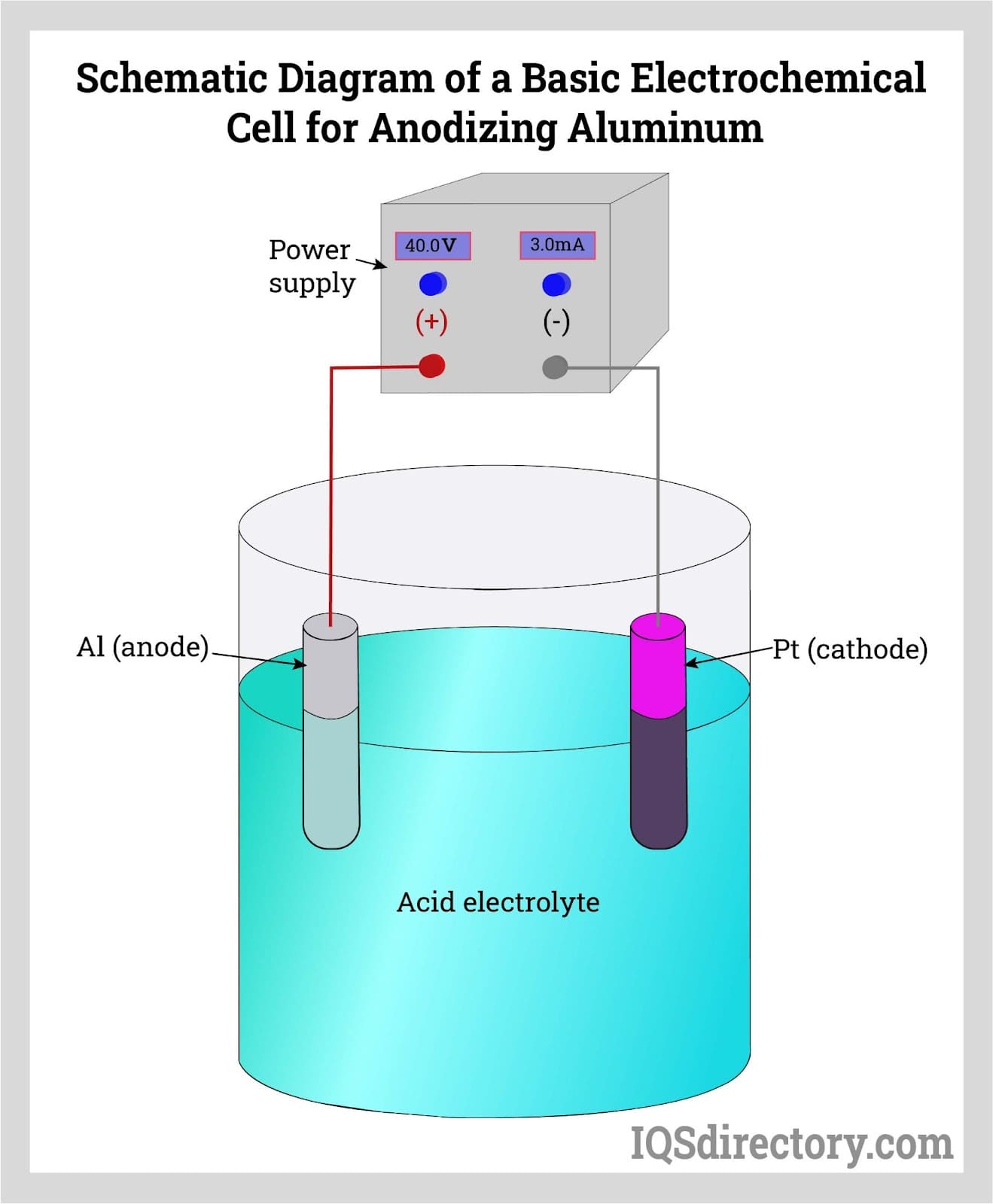 Anodizing is an electrolytic passivation process that increases the thickness of the natural oxide film on a metal surface, producing a durable, stable, and corrosion-resistant finish.
Anodizing is an electrolytic passivation process that increases the thickness of the natural oxide film on a metal surface, producing a durable, stable, and corrosion-resistant finish.
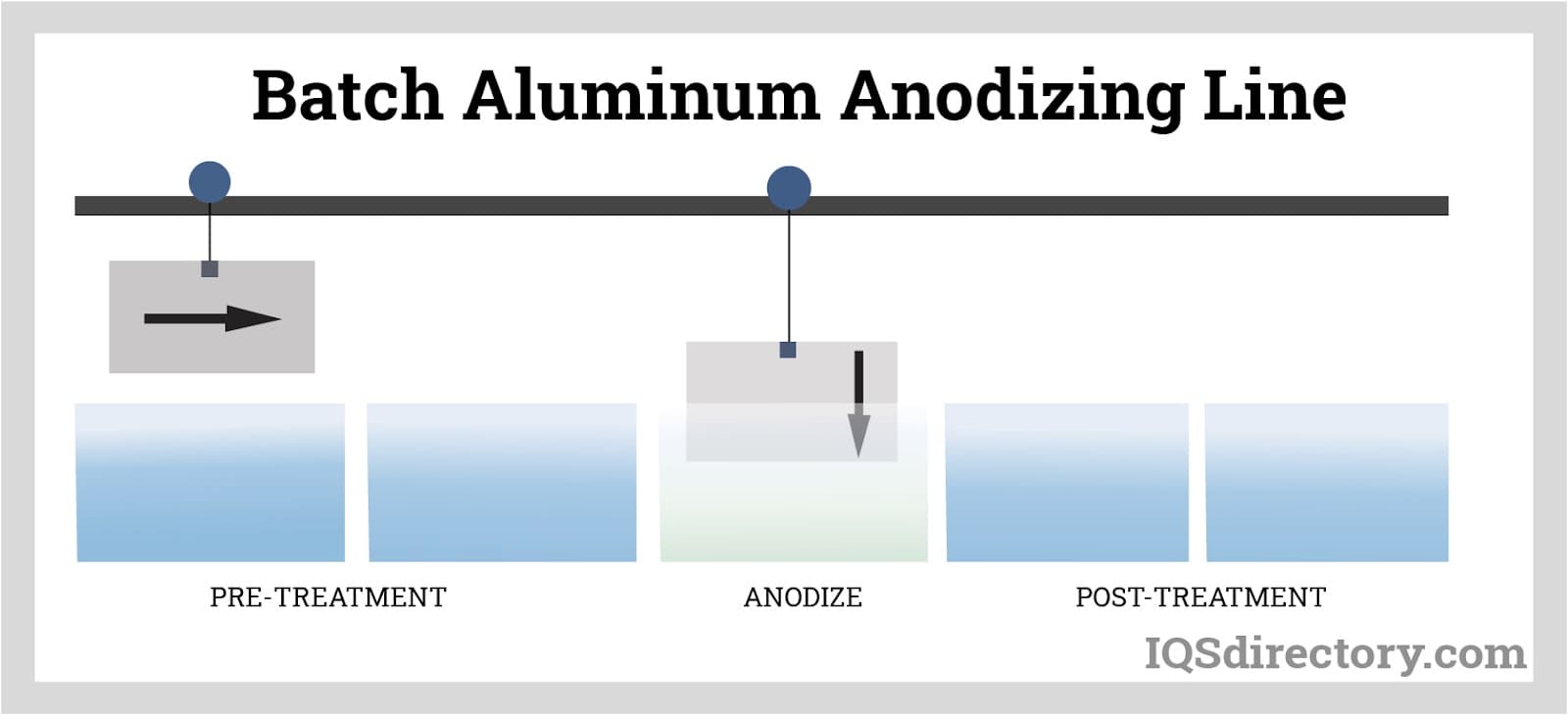 In batch anodizing, parts are mounted on racks and immersed through a sequence of process baths for cleaning, anodizing, coloring, sealing, and rinsing.
In batch anodizing, parts are mounted on racks and immersed through a sequence of process baths for cleaning, anodizing, coloring, sealing, and rinsing.
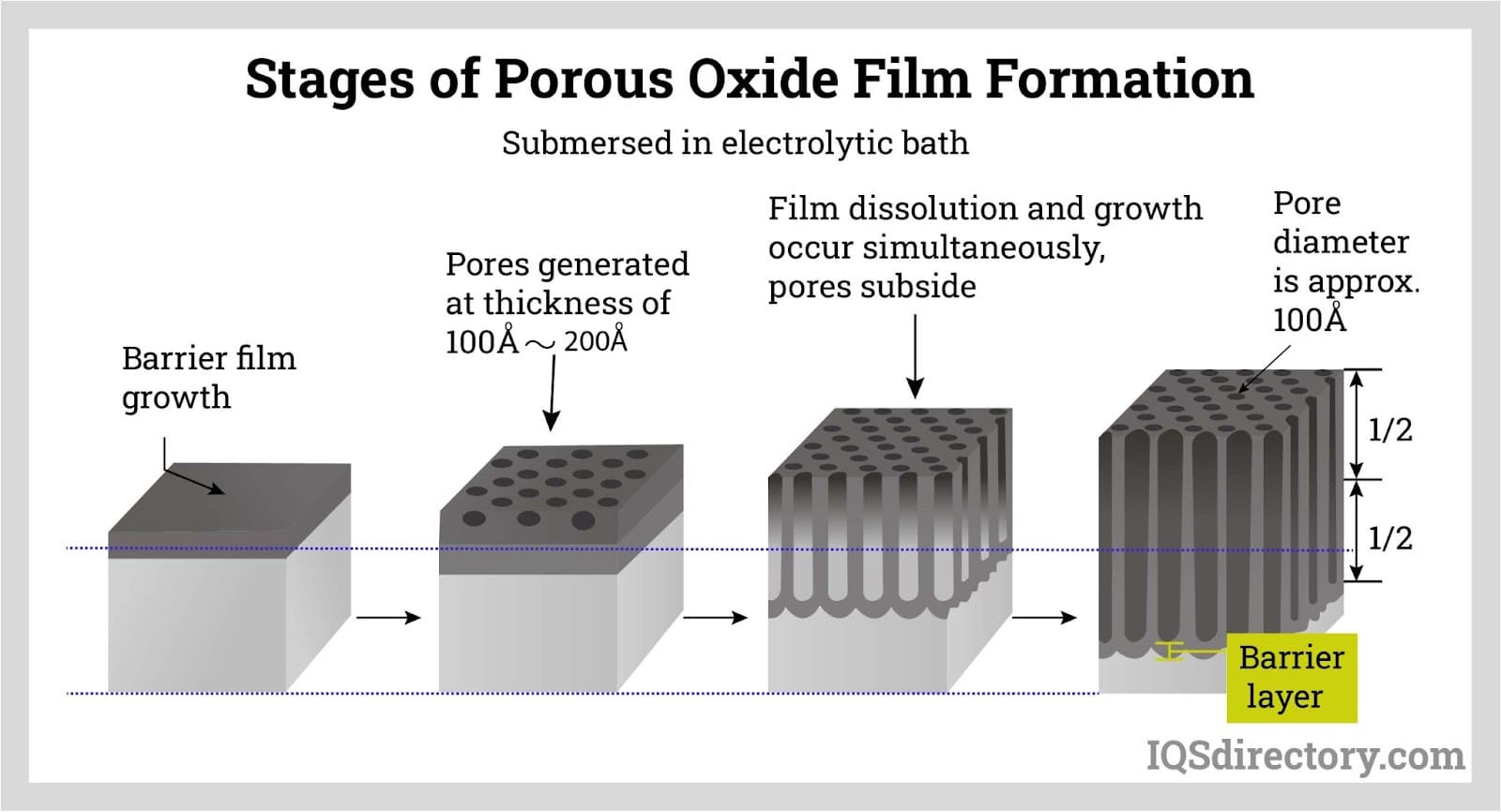 A porous oxide film forms in a dilute acidic solution, and its thickness generally changes with electrolysis time, voltage, and process conditions.
A porous oxide film forms in a dilute acidic solution, and its thickness generally changes with electrolysis time, voltage, and process conditions.
 Dyes or pigments fill the porous aluminum oxide layer created during anodizing, producing a durable finish that becomes part of the coating rather than a loose surface deposit.
Dyes or pigments fill the porous aluminum oxide layer created during anodizing, producing a durable finish that becomes part of the coating rather than a loose surface deposit.
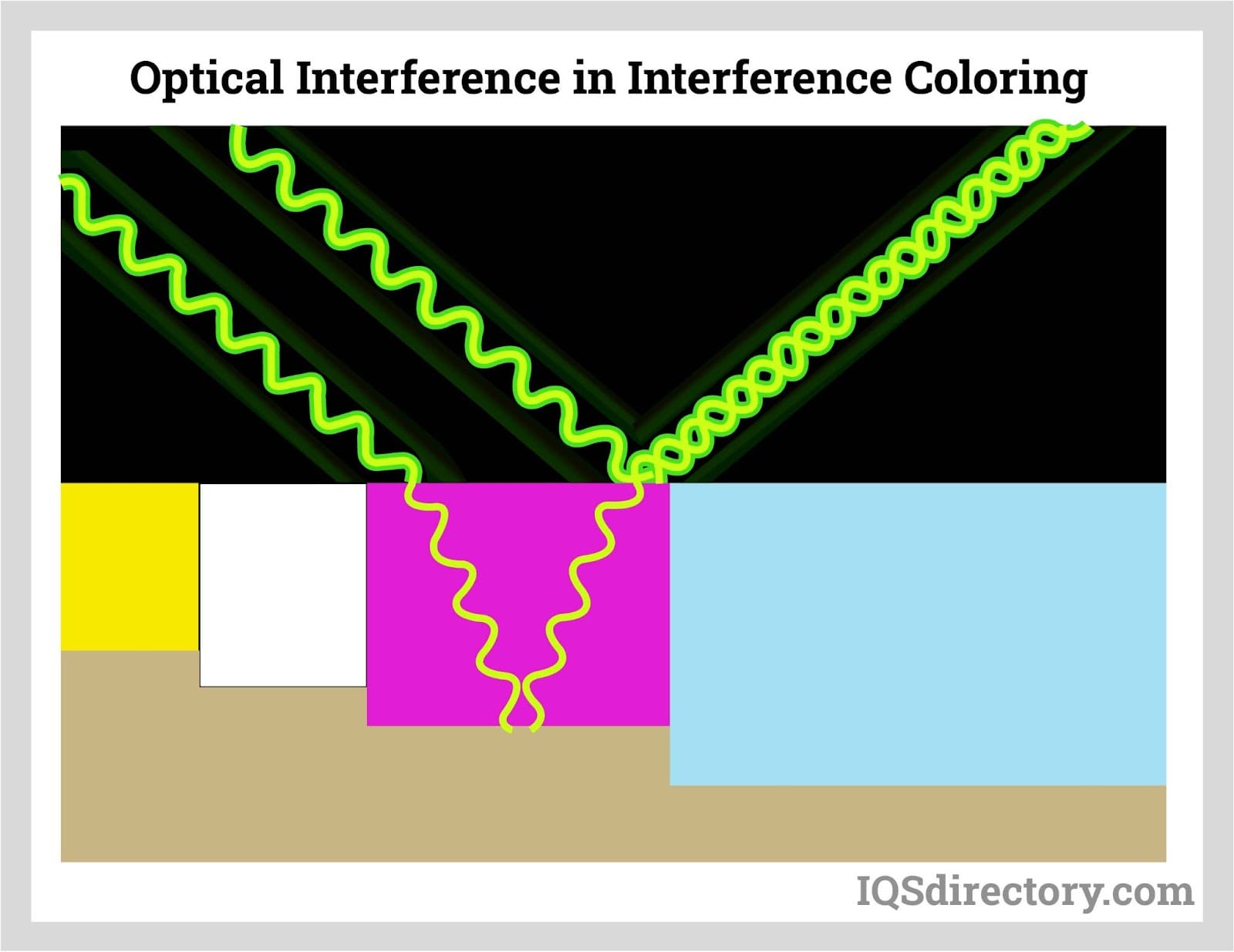 Interference coloring enlarges pore bases so metallic ions can be deposited electrolytically, producing lightfast decorative colors with strong visual depth.
Interference coloring enlarges pore bases so metallic ions can be deposited electrolytically, producing lightfast decorative colors with strong visual depth.
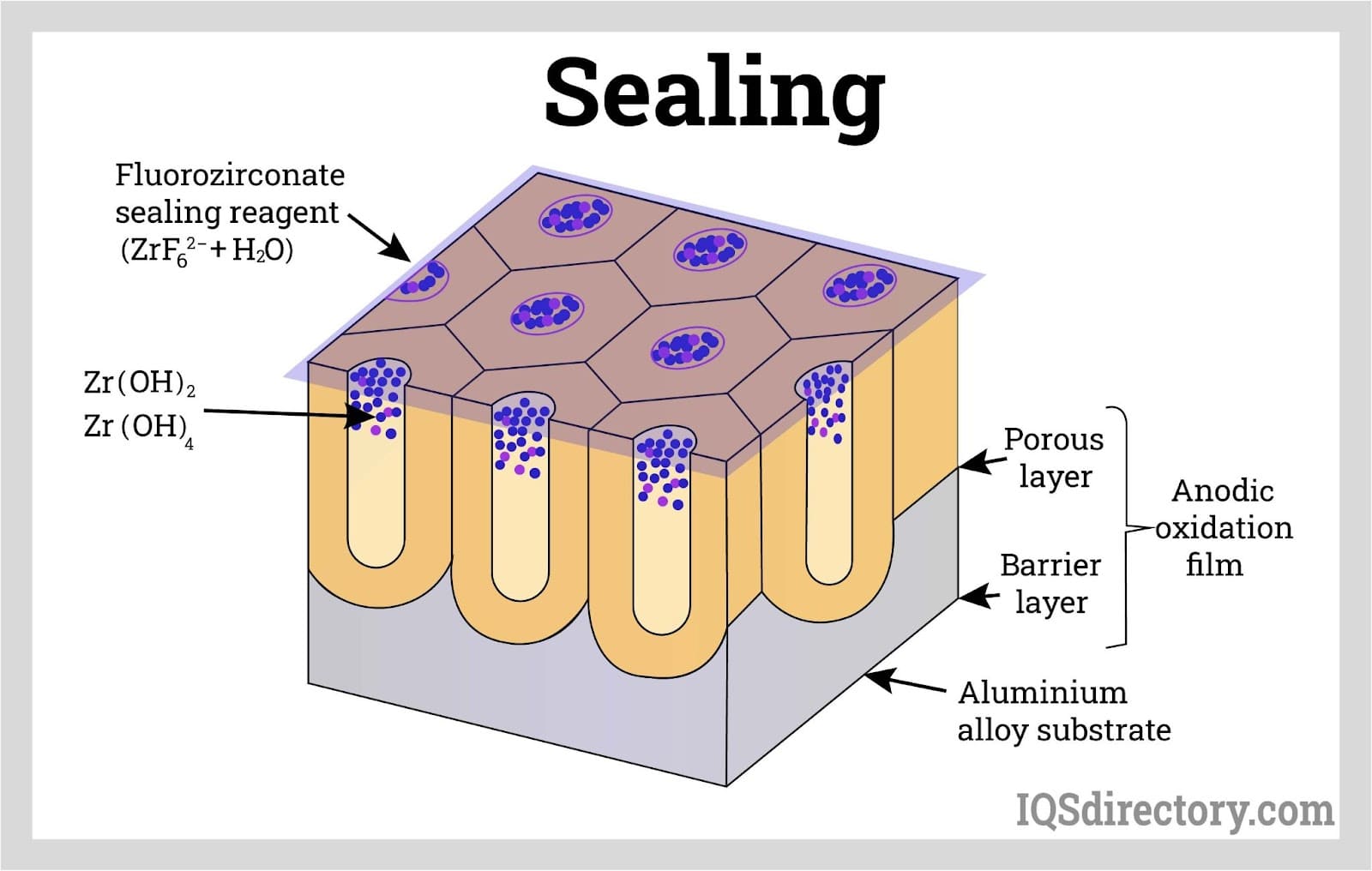 Sealing locks in dye, lubricant, or adhesive within the porous film and helps protect the anodized surface from corrosion, staining, and unwanted absorption.
Sealing locks in dye, lubricant, or adhesive within the porous film and helps protect the anodized surface from corrosion, staining, and unwanted absorption.
 Anodizing creates a stable aluminum oxide layer that is fully integrated with the underlying aluminum substrate for improved durability and finish performance.
Anodizing creates a stable aluminum oxide layer that is fully integrated with the underlying aluminum substrate for improved durability and finish performance.
Aluminum Anodizing Types
-
Anodizers
Service providers that perform anodizing to improve wear resistance, corrosion protection, finish durability, and surface appearance on aluminum and other metals.
Anodized Aluminum
Aluminum that has been processed electrolytically to improve corrosion resistance, wear performance, and decorative finish quality.
Anodized Metal
A metal treated through anodizing so that a controlled oxide layer forms on the surface for protection, color, or functional performance.
Anodizing
An electrochemical finishing process that converts the outer metal surface into a protective oxide coating.
Black Anodizing
A color anodizing process that produces a black dyed finish on anodized metal for decorative or functional use.
Bright Dip Anodizing
A pretreatment step in which metal is immersed in a chemical bath to create a brighter, more reflective surface before anodizing.
Chromate Conversion
A process used to improve corrosion resistance and create a conductive surface suitable for paints, powder coatings, and adhesives.
Clear Anodizing
A finishing process that produces a transparent or natural-looking anodized layer while preserving the base metal appearance.
Color Anodizing
The process of adding color to the anodized oxide layer before sealing.
Custom Anodizing
A specialized anodizing service tailored to part-specific needs such as hardness, color, coating thickness, sealing method, or end-use environment.
Hard Anodizing
A variation of anodizing that produces thicker, denser oxide films and is often selected for stronger wear resistance and surface hardness.
Sulfuric Acid Anodizing
A common anodizing method that forms a tightly bonded aluminum oxide coating offering corrosion resistance, visual appeal, and electrical non-conductivity.
Impregnation
The process of filling internal voids, cracks, or porosity in materials such as castings or powdered metal parts to prevent leakage or improve service life.
Titanium Anodizing
Titanium surface finishing used widely in jewelry and decorative applications because it provides attractive colors and good resistance for skin-contact uses.
Variations of the Aluminum Anodizing Process
Aluminum anodizing processes all follow the same basic principle: converting the metal surface into aluminum oxide through electrochemical action. Unlike paint, plating, or a simple applied coating, anodizing becomes part of the substrate itself, which is one reason it is widely used where finish adhesion and durability matter.
Although aluminum is the most common anodized metal, titanium, magnesium, and zinc can also be anodized for specialized performance or decorative needs. Niobium and tantalum are used as well, especially in jewelry and commemorative pieces, where oxide thickness and applied voltage influence final color.
Among anodizing candidates, aluminum remains the leading material because it combines low weight, good manufacturability, broad alloy availability, and strong response to the anodizing process. That makes it a top choice for industries seeking corrosion resistance, attractive finish options, and dependable long-term surface performance.
Aluminum Anodizing Companies and Suppliers
IQS Directory offers a broad listing of aluminum anodizing companies and suppliers. Buyers can use the directory to compare service providers, production capabilities, finish options, and application experience across the anodizing market.
The directory helps users find companies that provide aluminum anodizing services built around specific production goals, including decorative anodizing, hard coat anodizing, clear anodizing, and custom finishing. Company profiles may include website links, contact information, locations, videos, product details, and other business information that supports sourcing decisions.
Whether you are looking for anodizing services, process information, aluminum anodizing equipment, or custom anodized solutions, the directory is designed to help narrow options and connect buyers with qualified suppliers.
Aluminum Anodizing Terms
-
Abrasion
The wearing away of a surface through friction, rubbing, or repeated contact.
Activation
The process of making a metal surface chemically active before finishing or treatment.
Alloy
A material made from two or more elements, at least one of which is a metal, that exhibits metallic properties.
Alumina
An intermediate material derived from bauxite during aluminum production.
Anodizing Sheet
Aluminum sheet with metallurgical and surface characteristics suited for anodic oxidation and the production of protective or decorative films.
Bath
The chemical solution in which cleaning, anodizing, etching, coloring, sealing, or other finishing steps take place.
Brazing
A metal-joining process in which a molten filler metal flows between closely fitted parts.
Chemical Film
A chromate conversion coating applied to aluminum to improve corrosion resistance and support later finishing steps.
Corrosion
The gradual chemical or electrochemical breakdown of a material due to environmental exposure or reactive conditions.
Cryolite
A white mineral used in the production and refining of aluminum.
Deburring
The removal of burrs, fins, or sharp edges from metal parts through mechanical, chemical, or electrochemical methods.
Edging
The refining or shaping of metal edges through rolling, drawing, filing, or related forming methods.
Hardener
An alloy containing aluminum and at least one other metal that is used to modify molten aluminum.
Impurities
Unwanted elements present in aluminum compounds or alloys.
Inhibitor
A substance used to slow or reduce a chemical or electrochemical reaction, often to limit corrosion or control pickling action.
Melting Point
The temperature at which a metal changes from a solid to a liquid state.
Passivation
The process of making a chemically active metal surface more stable or less reactive.
Plating
A method of coating one material with a thin layer of another metal.
Pores
Microscopic openings in an anodized surface that can accept dyes or other materials before sealing while the coating remains dense and hard overall.
Rack
An electrically conductive holding fixture used to secure parts during anodizing and move them through process baths.
Refined Aluminum
Aluminum that has been purified to a high degree for manufacturing or finishing use.
Smelt
The process of melting or fusing ore to extract or purify the metal it contains.
Striation
Long or uneven lines caused by inconsistent coating, flow, or process conditions during anodizing.
Ultimate Strength
The maximum stress a material can withstand before failure.