Heat Treating
Heat treating refers to a range of processes used to alter the atomic structure of a metal or other crystalline material in order to impart beneficial physical, chemical, and mechanical properties such as increased strength or improved ductility. These techniques involve carefully controlled cycles of heating and cooling, which are used to change the internal microstructure of industrial materials. By exposing a material to extreme temperatures—either high or low—engineers can fundamentally adjust its internal structure, resulting in changes to the behavior and performance of the raw material or finished component.
It is important to distinguish between heat treating and hot forming, as the two are not the same. Hot forming involves the visible, plastic deformation of a material using heat, shaping it into a desired form. Heat treatment, by contrast, is aimed at creating internal, invisible atomic-level changes without altering the material’s overall shape or dimensions. Though closely related and often used in tandem—for instance, heat treating may follow immediately after hot forming—each process serves a distinct function in the manufacturing workflow. Keeping this distinction in mind is essential for understanding the role each plays in the development of a final product.
In modern manufacturing, heat treating is often used to improve hardness, toughness, machinability, fatigue resistance, wear life, dimensional stability, and overall part performance. Whether the goal is stress relief after welding, improved case hardness on a gear tooth, or better toughness in a tool steel component, heat treatment is one of the most widely used methods for tailoring a material to the demands of its end use. Buyers and engineers often search for heat treating services when they need a specific property outcome rather than a specific furnace cycle, which is why process knowledge and material expertise matter so much in this field.
Heat Treating FAQ
What is heat treating used for in metalworking?
Heat treating modifies the internal atomic structure of metals to enhance properties like strength, ductility, and wear resistance. It’s essential in manufacturing processes across industries such as automotive, aerospace, and construction.
How is heat treating different from hot forming?
Heat treating changes a material’s internal microstructure without altering its shape, while hot forming uses heat to reshape materials physically. The two are often used together but serve distinct purposes in production.
What are the main stages of the heat treating process?
Heat treating includes three stages: heating the material to a critical temperature, soaking it to ensure uniform heat distribution, and cooling it at a controlled rate to achieve the desired properties.
Which metals are commonly heat treated?
Steel is the most frequently heat treated metal, accounting for about 80% of all heat-treated materials. Aluminum, titanium, copper, and nickel are also commonly processed for enhanced mechanical performance.
What are the most common heat treatment methods?
Popular heat treatment methods include annealing, case hardening, normalizing, tempering, quenching, and nitriding. Each process targets specific material properties like hardness, ductility, or stress relief.
Why is temperature control critical in heat treating?
Precise temperature control ensures uniform heating and cooling, preventing cracks or distortion. Incorrect temperatures can compromise the material’s strength and lead to structural failure in use.
Where is heat treating commonly performed?
Heat treating is carried out in specialized facilities equipped with furnaces, quenching systems, and vacuum chambers. Many Midwest and Southern U.S. metalworking centers operate large-scale heat treatment plants for manufacturing and aerospace applications.
What safety precautions are important in heat treating?
Operators must use protective gear and maintain calibrated equipment to prevent burns, fires, or structural failures. Proper process control ensures safe, consistent results in industrial environments.
History of Steel and Heat Treating
Steel comprises the overwhelming majority of materials subjected to heat treatment, a fact that will be explored further in the following section. As a result, the history of heat treating is closely tied to the evolution of iron and steel production.
The origins of steel production can be traced back to the seventeenth century. Prior to that, metalworking relied primarily on cast iron—a high-carbon form of iron developed in Chinese and European blast furnaces. In Germany and England, a technique known as cementation emerged for increasing carbon content in molten pig iron. This method involved repeatedly heating wrought iron bars in powdered charcoal within sealed stone boxes. Cementation can thus be considered one of the earliest examples of deliberate heat treatment aimed at altering a metal’s internal composition.
Although steel was refined somewhat accidentally in the 1740s by English clockmaker Benjamin Huntsman, who improved its quality, the high cost of production kept it from becoming a mainstream industrial material. By the mid-nineteenth century, with the demands of the Industrial Revolution pushing the limits of iron production—particularly with the rise of railroads—Henry Bessemer introduced a more efficient method of reducing carbon in molten iron by injecting oxygen. While his method marked a major breakthrough, it took several decades of refinement before steel could be mass-produced affordably. Around 1860, German engineer Karl Siemens contributed to this progress by developing the open hearth process, using large, shallow furnaces to produce steel at an even greater scale. By the turn of the twentieth century, the open hearth process had become the backbone of global steel production.
Today, roughly two-thirds of all steel is produced using basic oxygen furnaces, which have replaced the open hearth method. It is estimated that the use of heat treatment adds between $15 and $20 billion annually in value to the metalworking industry.
That long connection between steelmaking and heat treating still shapes the industry today. Many of the most common commercial heat treatment practices were refined around the needs of steel because steel responds so strongly to changes in temperature, carbon content, cooling rate, and time at heat. Even now, many buyers use heat treatment not simply as a finishing step, but as a way to unlock the full potential of a steel grade after forging, machining, stamping, welding, or other forming operations.
Heat Treating Process
Typically, metallurgists or heat treatment specialists determine which processes are required to achieve the specific mechanical, chemical, and physical properties requested for a given application. Each material capable of undergoing heat treatment possesses a unique microstructure, made up of small grains or crystallites arranged in a complex lattice. This internal structure plays a crucial role in defining the characteristics of both raw materials and semi-finished products. While the science behind it is intricate, heat treatment can be broadly understood as a method of enhancing a material’s properties by adjusting the size and arrangement of its granular lattice.
The decision-making process behind selecting a heat treatment begins with analyzing the material itself. As noted, iron and steel—defined as iron alloys containing 0.2–2% carbon by weight—are the most commonly heat treated metals. Estimates suggest that up to 80% of all heat treated materials are steel. The wide variety of steel types is largely defined by the presence and proportion of carbon and other elements. For instance, plain steel contains only iron and carbon, while alloy steels include additional elements. Variants such as plain carbon steel, free cutting steel, chromium steel, and nickel-chromium steel are all categorized based on their carbon content and alloying elements. Steel may undergo heat treatment in its "raw" form straight from the mill, such as a bar, or after it has been formed through other methods like casting, forging, stamping, or extrusion.
While steel is the most common material for heat treatment, it is far from the only one. Other metals such as aluminum, titanium, copper , and nickel are also frequently heat treated. Moreover, heat treatment isn’t limited to metals—materials like ceramics, polymers, and certain glasses can also benefit from carefully applied heat processes.
Despite the range of available techniques, nearly all heat treatment processes follow the same three essential steps: heating, soaking, and cooling.
Heating Stage
This first step brings the material to a critical temperature—the point at which its internal lattice begins to change. The temperature reached, the rate of heating, and the duration all depend on the desired outcome. Because this stage initiates the transformation of the material’s structure, it has a major influence on the final properties of the product.
Soaking Stage
After the material reaches its critical temperature, it must be held there for a set period to allow heat to distribute evenly throughout. This stage is known as soaking. Soaking time can vary greatly—from just a few seconds to over sixty hours—depending on the material and the heat treatment goals.
Cooling Stage
Cooling, though involving a reduction in temperature, is just as integral to heat treatment as the heating phase. Cooling processes vary in speed and method. Rapid cooling, or quenching, uses gas or liquids to quickly draw heat from the material. Slower cooling methods may involve leaving the material in the furnace or exposing it to ambient air. Just like the heating stage, the cooling rate plays a critical role in determining the outcome. Faster cooling generally results in a finer grain structure, improving flexibility and ductility. Slower cooling tends to produce coarser grains, which enhances strength and rigidity. Together, these stages define the final properties of the heat treated material.
For many buyers, the process question is really a performance question. They may be trying to improve wear resistance on a part, reduce brittleness after hardening, restore machinability, relieve stress from fabrication, or achieve a hardened surface with a tougher core. That is why heat treatment providers are often valued not only for furnace capacity, but for their ability to connect heating profiles, soak time, atmosphere control, and cooling method to a precise property outcome.
Heat Treating Images, Diagrams and Visual Concepts
 During the stage of heating, the heat must even to prevent distortions or cracks.
During the stage of heating, the heat must even to prevent distortions or cracks.
 The soaking stage is used to keep the metal at an appropriate temperature to allow the metal to take the intended structure.
The soaking stage is used to keep the metal at an appropriate temperature to allow the metal to take the intended structure.
 The cooling stage allow the metal to be cooled to room temperature, which the rate of colling depends on the type of metal.
The cooling stage allow the metal to be cooled to room temperature, which the rate of colling depends on the type of metal.
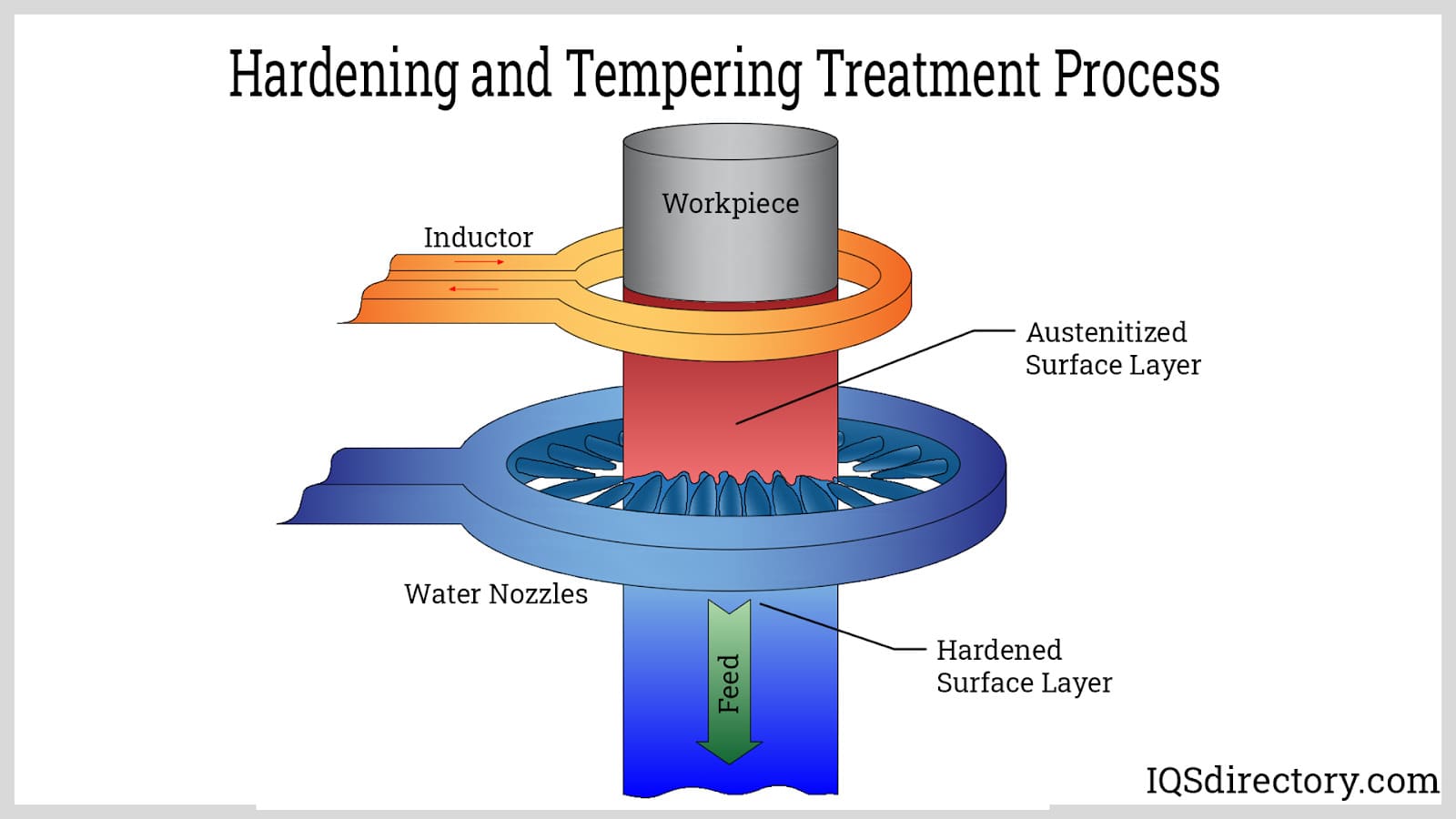 Hardening, a metal working process that occurs in a vacuum, while with tempering the metal is heated to high temperatures to increase its durability.
Hardening, a metal working process that occurs in a vacuum, while with tempering the metal is heated to high temperatures to increase its durability.
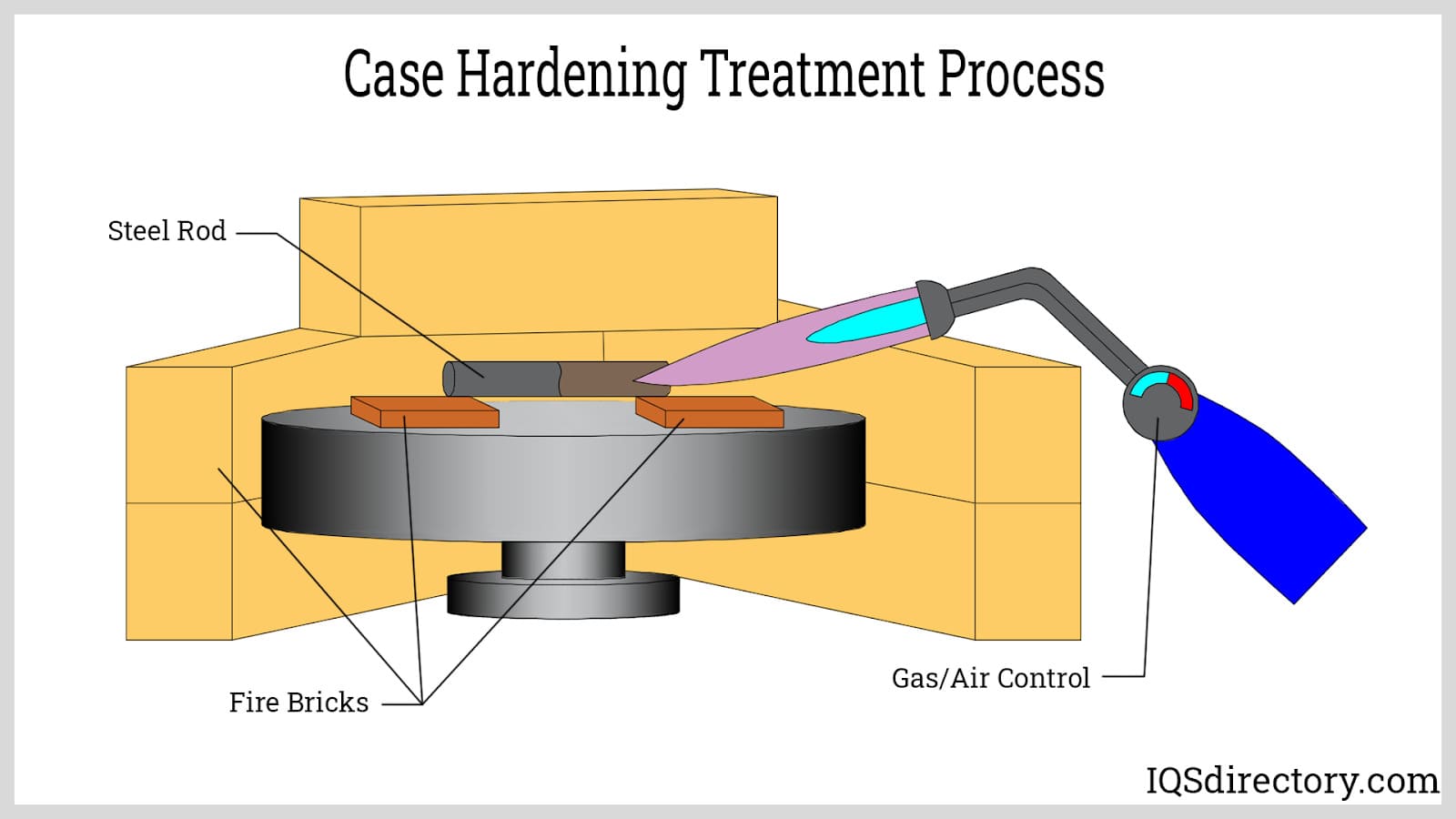 Case Hardening uses high heat with a combination of elements and chemicals to create a hardened outer layer.
Case Hardening uses high heat with a combination of elements and chemicals to create a hardened outer layer.
 Cyaniding treating deposits nitrogen and carbon atoms on the surface of the metal increasing the metals strength.
Cyaniding treating deposits nitrogen and carbon atoms on the surface of the metal increasing the metals strength.
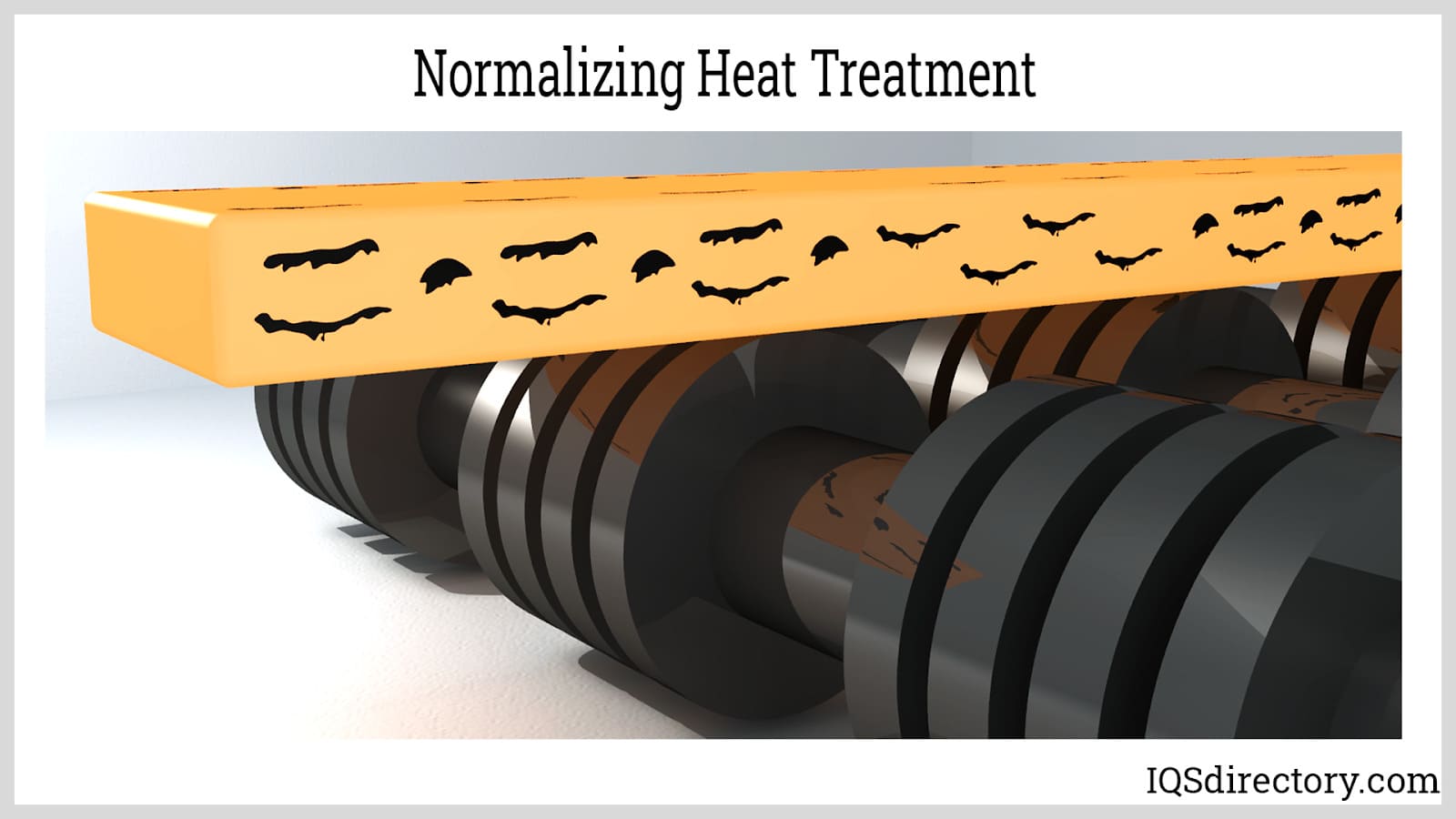 Normalizing removes the internal stresses after cold working and restores the structure of the metal.
Normalizing removes the internal stresses after cold working and restores the structure of the metal.
Types of Heat Treating
There are numerous methods of heat treatment, each producing distinct effects on a material's internal structure and performance. The main categories include annealing, case hardening, normalizing, and tempering. Additional techniques involve solution and aging processes, as well as specialized treatments for stainless steel, such as S3P (Specialty Stainless Steel Processes).
Annealing
One of the most commonly used heat treatments, annealing involves heating a material to a temperature above its critical point and holding it there for a set time, followed by slow cooling—often inside a furnace. This process reduces compositional segregation, enhances ductility, and allows the material to bend or form without cracking. It is particularly useful for preparing steel for cold-forming.
Case Hardening
This method focuses on chemical, rather than physical, transformation. Also known as surface hardening, it strengthens the outer layer of a part without affecting the core. Materials with high carbon or nitrogen content are heated, allowing those elements to migrate to the surface, forming a hardened shell. In contrast to annealing, case hardening increases rigidity and surface strength.
Normalizing
Considered a preliminary heat treatment step, normalizing is used to relieve stresses induced by earlier manufacturing processes like welding, forging, or cold rolling. The material is heated and then cooled in ambient air, effectively resetting any previous heat treatment history and preparing it for additional refinement.
Hardening and Quenching
This process is essentially the inverse of annealing. It involves heating the material to its transformation temperature and then rapidly cooling it—typically using water, brine, oil, polymer solution, or gas. While hardening refers to the heating phase, quenching refers to the rapid cooling. Quenching solidifies the structure but does not, by itself, change the microstructure unless paired with additional processes.
Tempering
Tempering follows hardening and reduces brittleness while enhancing toughness and ductility. It involves reheating the material—typically high-carbon steel—below its critical temperature. Low or mild carbon steels lack sufficient carbon for effective tempering. In some cases, cryogenic treatment using liquid nitrogen is used beforehand to further relieve stress.
Precipitation Hardening (Age Hardening)
This technique introduces an additional step after the initial heating and quenching. The material is reheated to a lower temperature to create a more uniform and refined microstructure. This process enhances strength and is commonly used in aluminum and superalloy components.
Nitriding and Other Processes
Methods like nitriding infuse nitrogen into the surface of a material to improve hardness, tensile strength, yield strength, and resistance to wear and fatigue. These techniques prepare materials for final use or for stock applications in demanding environments.
Austempering Method
A heat treatment process for steel involving quenching into a constant-temperature medium—typically a molten salt bath—to form bainite, a microstructure that improves strength and toughness.
Brazing Process
In this method, a molten filler metal bonds two base components at elevated temperatures. The result is a strong, often hermetically sealed joint.
Cyaniding Method
A surface hardening technique where steel is heated in contact with cyanide salts. The carbon and nitrogen absorbed into the surface form a hard shell, which is then quenched.
Flame Hardening
Used for localized hardening, this method involves heating specific surface areas with an acetylene torch followed by quenching. It’s suitable for components like gear teeth or camshafts.
Martempering Method
This involves quenching steel in a constant-temperature bath (such as salt) to reduce thermal shock, allowing the part to reach a uniform temperature before being cooled in air to form martensite. Tempering often follows.
Sinter Process (Sintering)
This technique bonds powdered materials into solid parts by heating them just below their melting point. Common in powder metallurgy, sintering is used across many industries for efficient, high-precision part production.
Tempering Process
Tempering ferrous metals after hardening reduces brittleness and increases toughness by reheating to a controlled temperature and then cooling again.
Vacuum Heat Treating
Conducted inside a vacuum furnace, this method eliminates contamination and oxidation during heating. The process is typically followed by quenching with inert gases and is often used for aerospace or high-precision components.
Because each method changes material behavior in a different way, heat treating is rarely a one-method topic. Many parts move through more than one thermal cycle before they are finished. A component may be normalized after fabrication, hardened and quenched for surface or through-hardness, and then tempered to restore toughness. In other cases, vacuum heat treating or atmosphere-controlled furnaces are selected to protect surface quality, reduce oxidation, or improve repeatability for more demanding industrial and aerospace work.
Applications and Products of Heat Treating
A wide range of industries regularly employ one or more heat treatment processes. In fact, within modern manufacturing, it is nearly impossible to operate without incorporating some form of heat treatment. Industries that rely heavily on these methods include automotive, hardware, construction, military, tool and die, stamping, trucking, shipping, aerospace, computing, and many others. Heat treatments are applied to an extensive variety of products, including cutting tools like saws and axes, gears, axles, fasteners, precision cutlery, dies, railroad components, and even musical instruments.
Heat treating is especially common wherever components must survive repeated loading, friction, impact, or temperature extremes. That includes gears, shafts, springs, bearings, dies, tooling, wear parts, stamped components, aerospace hardware, structural fasteners, and precision-machined parts. In many of these cases, the material’s usefulness depends not just on what alloy it is, but on whether it has been heat treated correctly for the intended service conditions.
Heat treatments require a variety of specialized tools to precisely control and alter the properties of materials. Chief among these are specialized furnaces, which are essential to the heat treating process and are available in many different configurations. Examples include critical furnaces, drop bottom furnaces, car bottom furnaces, elevator furnaces, turn-up furnaces, crucible furnaces, and age ovens. Some furnaces are designed specifically for induction or vacuum heat treating. In addition to these, tools such as lasers, torches, heating blankets, and salt baths are also employed. Each is carefully calibrated to modify the internal structure and composition of materials and components undergoing heat treatment.
The tool choice often depends on batch size, atmosphere requirements, temperature uniformity, part geometry, and the level of process control needed. High-volume commercial heat treating may rely on large production furnaces and automated quench systems, while specialized work may call for vacuum furnaces, induction systems, salt baths, or tightly controlled lab-style equipment. In every case, the equipment has to support consistent heating, repeatable soak time, and controlled cooling if the final material properties are going to be reliable.
Heat Treating Care and Safety
While heat treatments are highly effective, they also present a range of risks that must be managed with great care. Because these processes alter the physical strength and molecular stability of materials, it is critically important that all calculations are accurate and equipment is properly maintained. Malfunctioning or poorly calibrated tools can result in incorrect heating or cooling, potentially producing parts that fail under operational stress. Such failures can lead to serious consequences, especially in industrial settings where improperly treated materials may contribute to dangerous accidents.
Beyond the structural risks, the heat treating process itself poses physical hazards that must be addressed. Personnel involved in heat treatment operations should wear appropriate protective gear and have access to fire extinguishers to guard against burns, scalds, or fire outbreaks. In the end, strict adherence to equipment maintenance and process control allows heat treatment applications to safely and effectively serve a wide range of sectors—including industrial, commercial, and even domestic—by delivering materials tailored for enhanced strength and ductility.
Process discipline is a major part of heat treating safety and quality. Atmosphere control, furnace calibration, quench media condition, part spacing, and temperature verification all affect whether the finished component performs as intended. In high-demand industries, traceability and repeatability are just as important as the thermal cycle itself because even small deviations can affect hardness profile, distortion risk, and final mechanical properties.
Things to Consider When Choosing Heat Treating
When utilizing heat treated materials in any industry, it’s essential to understand that their performance capabilities depend heavily on the specific chemical composition of the base material. For instance, treating stainless steel. Because of these variations, it’s equally important to partner with a heat treating facility that offers a high level of expertise, experience, and well-maintained equipment. Key considerations should include the shop’s investment in modern technology and research, its consistency in results, and any relevant industry accreditations.
Heat treating providers are often brought in toward the final stages of a commercial project. Instead of specifying a particular process, clients usually communicate the required performance outcomes or functional characteristics of a part or material. As mentioned earlier, metallurgists and heat treatment professionals are then responsible for determining which treatment method best achieves those goals. For this reason, it’s critical to work with a heat treatment supplier known for reliability and exceptional customer service.
From the operational side, heat treatment providers must balance multiple considerations when establishing best practices—ranging from cost efficiency and product quality to environmental responsibility. Here are some examples:
Annealing remains one of the most widely used heat treatment methods. Still, in some situations, quenching may be preferred due to its generally lower cost.
The heat treating industry as a whole tends to be fragmented in its adoption of innovation and investment in R&D. While the initial cost of upgrading equipment and technology can be high, doing so often leads to long-term competitive advantages.
When choosing between quenching methods, environmental impact may guide decision-making. For example, while various substances can be used for quenching, polymer-based solutions are often the more environmentally friendly choice compared to traditional oil-based methods.
From a buyer’s perspective, choosing a heat treatment partner often means evaluating process capability, quality control, material knowledge, furnace capacity, turnaround, atmosphere options, distortion management, and the shop’s familiarity with the relevant industry standards. A strong provider is usually one that can translate a desired result like improved hardness, reduced residual stress, or better wear life into the right thermal process for the alloy and geometry involved.
Heat Treating Terms
-
Air Hardening Steel
A category of steel that hardens upon cooling in air, eliminating the need for quenching in water or oil.
Alloy
A combination of two or more metals designed to enhance specific properties such as strength, hardness, or melting point.
Austenite
A solid solution of iron and carbon formed when steel is heated above its critical temperature.
Bainite
A microstructure that forms when austenite is quenched and held at a constant intermediate temperature.
Carburizing
A heat treatment method that enriches the surface layer of steel with carbon by exposing it to a carbon-containing solid, liquid, or gas below its melting point.
Critical Temperature
The temperature threshold at which steel undergoes a structural transformation into austenite.
Decarburization
The depletion of carbon from the surface of a metal caused by high temperatures and exposure to reactive gases like air, oxygen, or hydrogen.
Hardenability
A metal’s capacity to harden uniformly when subjected to quenching.
Hardness
The ability of a material to resist deformation or surface penetration.
Impact Strength
The ability of a metal to absorb energy and resist cracking when subjected to sudden force or impact.
Martensite
A hard and brittle steel structure that forms when austenite is rapidly quenched.
Normalizing
A heat treatment that involves heating a metal above its critical temperature followed by air cooling to eliminate the effects of prior heat treatments or mechanical working.
Oxidation
A chemical reaction in which metal surfaces corrode due to exposure to atmospheric oxygen.
Pearlite
A layered microstructure of steel formed through the slow cooling of austenite in air.
Quenchant
The medium used to cool metals during quenching, including water, oil, brine, air, or molten salts.
Quenching
The rapid cooling of heated metal using a quenchant to modify its hardness and microstructure.
Temper
The condition or state of a metal, often adjusted through heat treatment to balance hardness and ductility.
Temper Color
The visible coloration on polished steel that appears during tempering, indicating the temperature the metal has reached.
Tensile Strength
The maximum stress a metal can withstand while being stretched before breaking.
Yield Strength
The stress point at which a material undergoes permanent deformation without breaking.