Nickel
Nickel (Ni) is a naturally occurring metallic chemical element. Its atomic number on the periodic table is 28 and its atomic weight is 58.71. Nickel plays an important role in healthy plant life, and it is also present in rock, soil, and many everyday food sources. Because of that broad presence, nickel is familiar to both industrial buyers and general readers researching corrosion-resistant metals, alloy materials, and high-performance manufacturing metals.
Because it is naturally occurring, it must be mined from deep within the earth instead of synthetically created in a lab. Called nickel ore, there are two main types of ore deposits: laterites, which are mainly composed of nickeliferous limonite and garnierite, and magmatic sulfide deposits, which are primarily composed of the ore mineral pentlandite. Understanding where nickel comes from helps buyers compare supply options, refining routes, purity levels, and the downstream performance of nickel metal and nickel alloy products.
In general, nickel has a silvery-white color, high toughness, is ferromagnetic and has excellent resistance to corrosion and rust. Some of its additional beneficial properties include its malleability, ductility, alloy-ability and high heat resistance—it has a melting point of 1453 degrees Celsius. Those traits explain why nickel is widely specified for stainless steel, plating, marine hardware, aerospace components, chemical processing equipment, battery technologies, electrical parts, and fabricated products that must hold up in demanding service conditions.
Nickel FAQs
What is nickel and where is it found?
Nickel is a naturally occurring metallic element with atomic number 28. It’s found in rocks, soil, and even plants, fruits, and nuts. Nickel must be mined from the earth, primarily from laterite and magmatic sulfide ore deposits, then processed into usable forms such as nickel sheet, nickel plate, nickel bar, nickel rod, tubing, plating solutions, and alloy feedstock for industrial manufacturing.
What makes nickel valuable in manufacturing?
Nickel offers exceptional toughness, corrosion resistance, and heat tolerance across a wide temperature range. It’s highly malleable, ductile, and alloyable, making it ideal for stainless steel production, plating, aerospace components, marine parts, electrical applications, and equipment exposed to chemicals, moisture, or repeated thermal cycling.
How is nickel extracted and refined?
Nickel is extracted through pyrometallurgical or hydrometallurgical processes. After refining, techniques like the Mond process can achieve very high purity, producing carbonyl nickel for advanced applications where chemistry control, cleanliness, and uniform material properties matter during fabrication, machining, coating, or alloy development.
What are the main nickel alloys and their uses?
Common nickel alloys include Monel®, Inconel®, and Kovar®. Monel resists seawater corrosion, Inconel performs well in high-heat service, and Kovar matches the expansion rate of glass in electronics manufacturing. Other nickel-based alloys are selected for heat exchangers, fasteners, seals, furnace parts, instrumentation, and aggressive chemical environments.
Where is most of the world’s nickel produced?
Nickel is produced in several major mining regions around the world, including Canada, Russia, Australia, Indonesia, and other established metals markets. For many buyers, supply availability, recycled content, mill lead times, alloy grade selection, and domestic processing options matter just as much as raw mine output when evaluating where to source nickel materials.
Why is nickel considered a sustainable material?
Nickel’s long service life and recyclability make it one of the world’s most reused industrial metals. Recycled nickel is widely used in stainless steel and other alloy systems, helping manufacturers reduce waste, manage material costs, and support long-term production goals without giving up corrosion resistance, durability, or high-temperature capability.
What products are made with nickel?
Nickel is used in stainless steel, coins, valves, batteries, and aerospace parts. It’s also found in household and industrial products such as kitchen sinks, cookware, process piping, heating elements, electronic devices, medical equipment, and fabricated components that need strength, conductivity, wear resistance, or long-term protection against corrosion.
Quick links to Nickel Information
The History of Nickel
Nickel has been used for thousands of years; we have traced back the use of nickel metal back to 3500 BC in Syria, when some of their bronzes contained at least 2% nickel. Other ancient accounts of nickel are found in Chinese manuscripts written between 1700 and 1400 BC, which describe "white copper" (cupronickel). It is possible that China was the source of cupronickel used in the 2nd century BC to mint coins for the Bactrian kings Agathocles, Euthydemus II and Pantaleon. Because nickel ore is easily mistaken for silver ore, and it was not officially identified and isolated as an element until 1751 by Swedish chemist Axel Cronstedt, people were using nickel-bearing materials long before they understood the metal scientifically.
Though nickel wasn’t officially discovered until the late 18th century, it got its name back in the 15th century. Its name comes from the Saxon term "Kupfernickel," which means "Devil’s copper." It earned this off-putting name from miners, who thought the metal, which is reddish-brown like copper, was too hard to mine and poisonous. Yes, the miners were being poisoned, but the arsenic in the ore was the real cause rather than the nickel itself. That early confusion is one reason nickel has such a distinctive history among industrial metals, nonferrous alloys, and mined material resources.
In the 19th century, the newly identified nickel rose to prominence in the form of plating and alloys like "nickel silver," which is an alloy of nickel, copper and zinc. It does not contain silver. Nickel then quickly gained popularity as coin material. The first coins with copper alloyed silver minted in the USA began circulating in 1857. In 1881, the Swiss government began circulating coins made from pure nickel. As manufacturing expanded, nickel became increasingly associated with corrosion-resistant finishes, wear-resistant surfaces, decorative metal finishing, and high-value alloy development.
Early the following century, scientists discovered stainless steels. Quickly, they found the alloy greatly benefited from the addition of nickel. This is because nickel adds high temperature resistance, corrosion resistance, toughness, and long-term durability. To this day, many grades of stainless steel contain nickel, and nickel alloys can now be found in chemical plants, jet engines, marine systems, power generation equipment, process vessels, and other demanding applications where long-term material performance matters.
Benefits of Nickel
Nickel is a great metal for many reasons. First, as compared to other metal materials, nickel offers more toughness, better corrosion resistance, a wider range of temperature resistance, and a variety of special electronic and magnetic properties. Second, nickel products have long working lives, with averages often ranging from decades when the material is matched properly to the application. In addition, nickel production can support efficient material use, and another aspect of nickel’s sustainability is its recyclability. Nickel and nickel alloys are among the most heavily recycled materials in the world. In fact, it is estimated that a large share of the nickel content of many modern stainless steel products comes from recycled materials.
Nickel also has far reaching socio-economic benefits. For example, many important elements of human life are dependent on the performance of nickel-containing products. Such elements include clean drinking water, food, power and heat, medicines, transportation, electronics, and industrial processing systems. Often, nickel is the material buyers turn to when they need a balance of corrosion protection, strength, fabricability, and long service life. In addition, nickel is known as an enabling technology that helps users create new industries, products, jobs and benefits while delivering improved performance in a wide range of sectors. If you are comparing nickel alloys for marine hardware, chemical handling, battery components, or heat-resistant equipment, those performance traits usually shape the final purchasing decision.
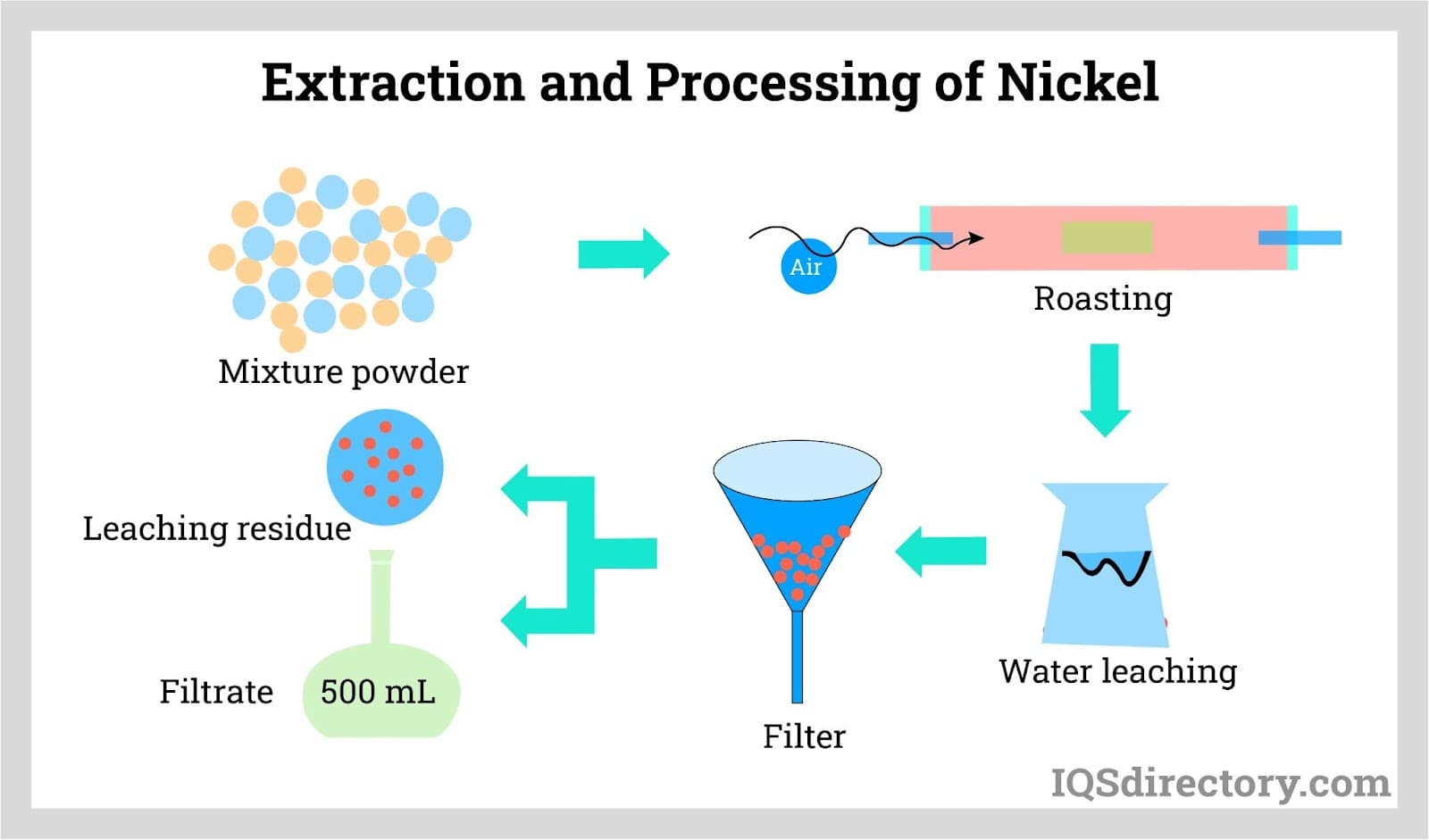
Production Process of Nickel
- Extraction Process
- Nickel ore is extracted from deposits through the process of extractive metallurgy, in which the raw metal material is extracted from the ore and purified into a refined form. The most common way the raw material is extracted is through pyrometallurgical extraction, but recent developments have made hydrometallurgy another viable process depending on ore chemistry, recovery goals, operating cost, and the desired downstream nickel product.
- Nickel Processing and Refinement
- Using conventional roasting and reduction processes, the refined form of nickel is able to yield a purity of greater than 75%. However, this level of purity can be increased through further processing. The Mond process, in which nickel oxides (nickel combined with oxygen) are converted into purified nickel, is able to achieve a purity of 99%. The nickel produced during this process is known as carbonyl nickel because carbon monoxide is utilized in the process, making it useful for applications that require tightly controlled chemistry.
- Shaping Process
- After nickel has been refined, nickel suppliers shape the metal into various shapes and parts for fabricators to use down the line. Nickel suppliers like to have in stock nickel shapes such as nickel sheet, nickel plate, nickel rods, nickel bar and, sometimes, nickel tubing. Stock availability, gauge range, bar dimensions, and alloy grade can all influence which supplier is the best fit for a given order.
- To create these shapes, nickel suppliers use a wide variety of fabrication processes, such as extrusion, cold drawing or electroplating. Depending on the end use, manufacturers may also incorporate rolling, machining, cutting, finishing, and secondary processing to achieve the desired tolerances, surface finish, and mechanical properties.
- Nickel Extrusion
- During nickel extrusion, nickel is heated to a molten or highly workable state and then shaped by forcing the pliable nickel through a die. Nickel extrusion can produce nickel sheets, bars, rods and tubing. Note: Nickel extrusion is the least common form of nickel forming, but it can still be a useful option when buyers need custom cross-sections or specialized profiles.
- Cold Drawing
- A more common process of shaping nickel is cold drawing. This is used to make tubes and wire. In cold drawing, no heat is added to the process, but the nickel tube or wire is drawn through a series of smaller and smaller dies, in order to reduce its diameter. This method can also help improve dimensional consistency and surface finish for precision applications.
- Electroless Nickel Plating
- The most common way to form nickel is through electroless nickel plating. During electroless nickel plating, a catalytic reduction process of nickel ions occurs in an aqueous solution that contains a chemical reducing agent. As a result, nickel metal is deposited without requiring electrical energy. The main cause of the deposition of nickel ions as well as their reduction is the chemical reducing agent within the solution. In addition, the nickel ion deposits are very uniform in thickness in terms of both shape and size because the driving potential of the chemical reducing agent is constant at all points of the surface of the component, as long as the agitation is enough to ensure a uniform concentration of nickel ions and the reducing agent. That uniformity is one reason buyers often choose electroless nickel plating for wear resistance, corrosion protection, dimensional control, and surface hardness.
- Alloys and Compounds Made
- One of the transition elements, nickel is often combined with other elements in order to form various alloys and compounds. Typical alloying materials include iron, chromium, copper, cobalt, aluminum and more. Iron-nickel (or nickel-iron) and nickel-chromium alloys are among the most popular, thanks to the strength that both iron and chromium lend. The most commonly formed nickel alloy is stainless steel, which is a nickel-chromium alloy composed of mostly iron and approximately 18% chromium and 8% nickel.
- Nickel is also used to make various other metal alloys including nickel 200 (which is made of 99.6% nickel), Invar® (a nickel-iron alloy), Monel®, Kovar®, Nichrome®, Inconel® and the various Hastelloys®. These materials are selected when buyers need a combination of strength, thermal stability, oxidation resistance, low expansion, magnetic behavior, or chemical resistance that standard steels often cannot provide.
- Monel®
- The general name for a series of nickel alloys that have a composition of nickel (approximately 67%), copper, iron and minute amounts of other elements. Although this nickel-copper alloy is very difficult to machine because it instantly hardens in reaction heat, it has incredible corrosion resistance. For this reason, it is often used in marine applications, such as trolling wire, pump shafts, seawater valves, and other saltwater handling equipment.
- Kovar®
- A term that encompasses a wide variety of nickel alloys that are compatible with the thermal expansion characteristics of borosilicate glass. Kovar® is mainly composed of nickel and cobalt, with minute amounts of silicon, manganese and copper, and is a ferrous alloy. Commonly used in electronics applications, Kovar® is often used as electroplated conductors for parts such as vacuum tubes, x-ray tubes, hermetic seals, and glass-to-metal assemblies.
- Inconel®
- Also referring to a family of nickel alloys, this nickel chromium alloy is actually a super alloy, meaning that it is a high-performance alloy. Exhibiting exceptional heat-resistance, Inconel® is composed mainly of nickel, chromium, molybdenum and niobium, with trace amounts of cobalt, manganese, copper, aluminum, titanium, silicon, carbon, sulfur, phosphorus and boron. It is regularly specified for aerospace, turbine, furnace, exhaust, and high-temperature process equipment where oxidation resistance and mechanical strength must remain reliable.
Nickel Images, Diagrams and Visual Concepts
 Nickel processed into a usable material.
Nickel processed into a usable material.
 Extractive metallurgy, a process used to extract the raw metal from its ore.
Extractive metallurgy, a process used to extract the raw metal from its ore.
 Inconel alloys contain nickel and chromium making them resistant to corrosion, oxidation, and elevated service temperatures.
Inconel alloys contain nickel and chromium making them resistant to corrosion, oxidation, and elevated service temperatures.
 Permalloy is a nickel alloy that contains 80% nickel and 20% iron, which makes the metal highly magnetic for specialized applications.
Permalloy is a nickel alloy that contains 80% nickel and 20% iron, which makes the metal highly magnetic for specialized applications.
 A nickel-based alloy known for resistance to corrosive acids and other aggressive processing environments.
A nickel-based alloy known for resistance to corrosive acids and other aggressive processing environments.
 This alloy contains iron, nickel, cobalt and trace amounts of manganese, silicon, and carbon, allowing it to have a low coefficient of thermal expansion.
This alloy contains iron, nickel, cobalt and trace amounts of manganese, silicon, and carbon, allowing it to have a low coefficient of thermal expansion.
 This alloy has corrosion resistance, malleability, a low coefficient of thermal expansion, and high strength for demanding service environments.
This alloy has corrosion resistance, malleability, a low coefficient of thermal expansion, and high strength for demanding service environments.
 The name for wrought nickel that has a purity of about 99.6%, often chosen when high-purity nickel is required.
The name for wrought nickel that has a purity of about 99.6%, often chosen when high-purity nickel is required.
Nickel Types
- Brushed Nickel
- A finish created by a rough surface going over the metal to create very small patterned lines, making a distinctive look, yet retaining its metallic luster. It is common in architectural hardware, fixtures, and decorative components where appearance and durability are both important.
- Casting Alloys
- Alloys used to form objects in molds, and they are more easily molded than other alloys. They are often selected for complex shapes, repeatable geometry, and industrial parts that benefit from near-net-shape manufacturing.
- Cupro Nickel
- A nickel alloy made up of nickel, copper, iron and manganese (or other strengthening impurities). It does not corrode in seawater and is used in various marine applications such as piping, condenser tubing, desalination equipment, and offshore systems.
- Inconel Alloy
- A nickel-base alloy, but also has chromium and iron, and it is used in gas turbine blades, exhaust systems, and other high-temperature equipment.
- Invar Alloy
- A trademark alloy of nickel and iron and is usually used in tuning forks, measuring tapes and other instruments where low thermal expansion is desired.
- Kovar
- A registered trademark that refers to a low expansion alloy that is composed of iron, nickel, cobalt and trace amounts of the elements manganese, silicone and carbon.
- Monel
- A trademark alloy made up of mostly nickel, as well as copper, iron and other trace elements. It cannot be easily corroded by many acids, and it can withstand harsh service conditions, but it is hard to machine because it hardens instantly.
- Nichrome
- A nickel-chromium alloy used for resistance heating elements because it can withstand high temperatures and has a high electrical resistance.
- Nickel 200
- Refers to wrought nickel that is commercially pure (99.6%) and is often selected for chemical handling, caustic service, and fabricated parts where purity and corrosion resistance are valued.
- Nickel Alloys
- Alloys made up of more nickel than anything else and designed for heat resistance, corrosion control, strength, magnetic performance, or specialized thermal properties.
- Nickel Bars and Rods
- Straight, solid products of nickel or nickel alloys that can be extruded. These products can have a variety of shapes, circular, triangular, square and more. Nickel rod refers to a solid, straight bar that can be either round or square, although round is much more common, and is composed of either high purity nickel or nickel-based alloys for machining and fabrication.
- Nickel Metal
- An element listed on the periodic table that is silver in color and both ductile and malleable, widely used as a base material for alloys, coatings, electrical products, and corrosion-resistant components.
- Nickel Plates
- Consist of rolled nickel and are used as components in industrial fabrications, processing equipment, and structural applications where alloy performance is required.
- Nickel Pricing
- Usually only quoted for a short period—usually less than a week long—as the price of nickel alters regularly with market conditions, alloy demand, supply chain movement, and mill availability.
- Nickel Sheet
- A flat plane composed of nickel or nickel alloys and commonly specified for fabrication, stamping, chemical processing equipment, and applications that require formed corrosion-resistant material.
- Nickel Suppliers
- Provide high purity nickel and nickel alloys, which share characteristics such as malleability, somewhat ferromagnetic behavior, hardness, ductility, and electrical and heat-conductivity, to diverse companies for further processing, fabrication, machining, or plating support.
- Nickel Tubing
- A hollow, cylindrical or rectangular rod that can be used as equipment components or to transport fluids or gases in chemical, marine, energy, and process systems.
- Permalloy
- An 80/20 alloy of nickel and iron which is easily demagnetized and magnetized, making it useful for magnetic shielding, transformers, and precision electronic applications.
Nickel Applications
Nickel is valued for its positive properties, detailed in the section above. It is used to make products and both decorative and functional coatings. It is also used extensively to make alloys, which are in turn used to make products of all kinds. Buyers researching nickel applications often compare corrosion resistance, high-temperature performance, machinability, magnetic behavior, conductivity, and total lifecycle value before choosing a grade or alloy family.
Since nickel can be found in a wide range of metals, it is utilized in a correspondingly vast number of industries including currency and coinage, consumer products, healthcare, chemical, industrial, food and beverage, electronics, military, transport, aerospace, architecture and marine. What nickel alloy is right for your application? The answer usually depends on temperature exposure, chemical contact, fabrication method, required strength, and whether the part must resist oxidation, saltwater, wear, or repeated thermal cycling.
Nickel Products Produced
Nickel and nickel alloys are used to make a wide variety of products. Approximately 65% of all nickel produced is used to manufacture stainless steel products, while another 20% is used to make other non-ferrous and steel alloys. Still another 9% is used to make plating, while about 6% is used to make other products, such as coins (including the American nickel and the Euro), fittings and valves, electronic devices, electric guitar strings, microphone capsules, rechargeable batteries for portable equipment and hybrid cars, some light bulbs and microwave tubes, rocket motor cases, missile components, food processing equipment, pots and pans, kitchen sinks and medical equipment. Nickel-containing materials are also widely associated with chemical handling systems, marine hardware, fabricated process components, and other demanding industrial products where long service life matters.
Nickel Grades
| Material |
Tensile Strength at Break (MPa) |
Tensile Strength, Yield (MPa) |
Modulus of Elasticity (ksi) |
| Pure Nickel |
45.0 - 317 |
59 |
30000 |
| All Nickel Alloys |
45.0 - 2070 |
59.0 - 4830 |
4060 - 34100 |
| Nitinol - High-Temperature Phase |
754 - 960 |
560 |
10900 |
| Nitinol - Low-Temperature Phase |
754 - 960 |
100 |
4060 |
| Inconel |
621 - 1550 |
195 - 1390 |
25700 - 32100 |
| Kovar |
517 |
345 |
20000 |
| Monel |
385 - 1100 |
134 - 790 |
~24500 |
| Nickel 200 |
379 |
103 |
10900 - 28000 |
*These figures are guidelines based on industry research; they should not be presumed accurate under all circumstances and are not a substitute for certified measurements. The information is not to be interpreted as absolute material properties nor does it constitute a representation or warranty for which we assume legal liability. User shall determine suitability of the material for the intended use and assumes all risk and liability whatsoever in connection therewith.
Overseas Nickel Market
Nickel ore deposits are widely distributed across major global mining regions, and international supply continues to shape pricing, availability, and sourcing strategy for manufacturers. Buyers often evaluate recycled content, domestic inventory, mill lead times, alloy conversion capability, and import logistics when reviewing overseas nickel market options. Because nickel is heavily recycled, part of the supply burden can be reduced while also supporting more stable material availability for stainless steel, plating, and specialty alloy production. For many purchasers, the best sourcing decision comes down to reliability, grade availability, order size, and service support rather than geography alone.
Things to Consider When Purchasing Nickel
It is easy to see why you’d want to purchase nickel material. Unfortunately, it’s less easy to know what type of nickel material best fits the job. For the best results, we recommend you reach out to a supplier that will guide you through the market offerings, stock forms, alloy families, and fabrication options. To that end, we’ve provided you with a list near the top of this page of capable and reliable nickel manufacturers and suppliers. Get a feel for who they are by browsing their respective webpages. To figure out which one is right for you, pick out three or four you find most promising, then reach out to them with your application details and requirements. Compare and contrast their answers. Pay attention not only to prices, but also secondary services, lead times, delivery policies, material certifications, and overall customer service. If you are asking questions such as which nickel grade handles heat best, which alloy resists seawater, or which product form is easiest to fabricate, those discussions can quickly narrow the field and help you choose the right manufacturer.
Nickel Terms
- Alloy
- A combination of two or more metallic elements that are usually dissolved into each other or fused together.
- Cold Forming
- Deformation of a metal at a low enough temperature to prevent re-crystallization during cooling.
- Conductivity
- A metal’s ability to conduct electricity. Nickel is a good conductor, and therefore is used in wires, electronic assemblies, and other electrical applications.
- Ductility
- The capability of a metal, such as nickel, to allow deformation or shaping before finally fracturing.
- Electroless Nickel Plating
- A process in which nickel coating is applied to a surface in a controlled chemical reduction. Electrons used are not supplied electrically, but by a chemical reducing agent.
- Electroplating
- A process by which metal ions are attracted to a solid metal electrode. As the ions bind to the surface of the metal, they become a thin coating, which forms a protective layer to prevent corrosion and wear.
- Extractive Metallurgy
- The process of purifying and recycling metal that was extracted from ore.
- Ferromagnetic
- It is the most familiar form of magnetism. Permanent magnets are ferromagnetic, and so are the metals that are attracted to them, such as nickel.
- Hydride
- Any binary compound of hydrogen and another element.
- Malleable
- The characteristic of some metals, meaning they have the ability to be shaped or formed by applying pressure.
- Non-ferrous
- A type of metal which does not contain iron.
- Oxidation
- The reaction in which oxygen is added and causes the removal of electrons from the reactant.
- Superalloy
- An alloy with a base element of nickel, nickel-iron or cobalt, which has corrosion resistance, ability to withstand high temperatures, mechanical strength and good surface stability.