Tungsten
Tungsten stands as one of the most remarkable naturally occurring metallic elements, bearing atomic number 74 and frequently known by its alternative name wolfram. Derived primarily from wolframite ore—which directly inspired its periodic table symbol W—tungsten is also sourced from scheelite deposits. This dense metal exhibits a distinctive color palette ranging from steel gray to tin white. Tungsten distinguishes itself through exceptional tensile strength, superior density, and unparalleled resistance to extreme heat. It possesses the highest melting point of any pure metal at 6192 °F and maintains the lowest vapor pressure, while ranking as the heaviest element suitable for practical industrial applications.
Tungsten FAQ
What makes tungsten different from other metals?
Tungsten stands out for its extremely high melting point, exceptional strength, and density. It has the lowest vapor pressure among metals and maintains hardness even at high temperatures, making it ideal for industrial, aerospace, and electrical applications.
Where is most tungsten mined today?
Most tungsten is mined in China and Russia, with additional production from Portugal, Austria, Peru, and Bolivia. The United States imports tungsten due to minimal domestic mining, relying instead on recycling and overseas supply chains.
How is tungsten produced and processed?
Manufacturers typically sinter or mold tungsten into billets before shaping them into rods, plates, foils, or wires through drawing, grinding, and die cutting. Finishing processes include cleaning or precision grinding for specific applications.
What are the main types of tungsten materials?
Tungsten materials include tungsten carbide, cemented carbide, alloyed tungsten, heavy-metal tungsten alloys, pure tungsten, and tungsten-based chemicals. Each type differs in hardness, conductivity, and strength for various industrial uses.
What industries rely on tungsten products?
Tungsten is vital in aerospace, construction, mining, semiconductors, electronics, and medical sectors. Its strength and heat resistance make it essential for cutting tools, electrical contacts, welding electrodes, and lighting components.
Why is tungsten considered environmentally beneficial?
Tungsten is low in toxicity and highly recyclable. Recycling reduces production costs and environmental impact, making it an eco-friendly choice for U.S. manufacturers and global industries seeking sustainable materials.
What should companies consider when choosing a tungsten supplier?
Buyers should select experienced suppliers who meet their budget, timeline, and quality needs. U.S.-based manufacturers often provide faster delivery, easier communication, and compliance with domestic regulations.
Benefits of Tungsten
Tungsten delivers a compelling combination of performance characteristics that make it indispensable across demanding sectors. Its inherently low toxicity profile supports safer handling for workers and reduced environmental burden compared to many alternative metals. The material’s high recyclability further enhances its appeal, enabling repeated reuse that lowers overall production expenses while decreasing reliance on primary mining operations. In terms of mechanical properties, tungsten exhibits outstanding strength; when combined with carbon to form tungsten carbide, its hardness approaches that of diamond. Even in pure form, tungsten rivals titanium for structural integrity under extreme conditions, delivering reliable performance where other materials would soften or degrade.
Additional advantages include excellent thermal and electrical conductivity paired with superior corrosion resistance. These attributes support long service life in high-temperature environments and electrical systems. Decision-makers evaluating tungsten materials frequently explore questions such as “What benefits does tungsten offer over stainless steel or titanium in high-heat applications?” or “How does the recyclability of tungsten impact long-term project costs?” Addressing these considerations helps align material selection with both performance requirements and sustainability goals.
The History of Tungsten
Early encounters with tungsten trace back to the 1500s among tin miners in the Erz Mountains along the border of present-day Germany and the Czech Republic. While processing tin ore, workers encountered a problematic mineral that produced stubborn slag and complicated extraction. They named it “wolfrahm,” translating to “wolf froth,” because the substance appeared to devour the tin like a wolf while forming a foamy layer during smelting. Its dark, fibrous appearance reinforced this vivid description. In 1546, Georgius Agricola documented the mineral in his influential work De Natura Fossilium, labeling it “spuma lupi”—Latin for wolf froth or wolf cream.
- During the 1700’s
- Tungsten derives its common name from the Swedish term for another ore, scheelite, known locally as “tung sten” or heavy stone. In 1758, Swedish chemist Axel Fredrik Cronstedt identified scheelite and suspected it contained a new element, though isolation proved elusive at the time. Progress continued when fellow Swede Carl Wilhelm Scheele successfully isolated the oxide around 1781. Finally, in 1783, Spanish chemists Fausto and Juan José de Elhuyar y de Suvisa reduced wolframite ore to produce the pure metal, marking tungsten’s formal discovery.
- During the 1800’s
- Industrial interest grew as researchers experimented with tungsten steel starting in 1855, with the first patents granted in 1858. Early tungsten steels proved costly, limiting widespread adoption. By the later decades of the century, however, tungsten’s value for hardening and alloying steel became clear, leading to the creation of high-speed tool steels. These advanced materials gained international recognition at the 1900 World Exhibition in Paris and continue to serve manufacturing needs today.
- During the 1900’s
- In 1903, American engineer W.D. Coolidge achieved a breakthrough by developing ductile tungsten wire through powder metallurgy techniques. He doped tungsten oxide to create metal powder, then pressed, sintered, and forged it into tungsten rods before drawing fine filaments. This innovation enabled European inventors to produce the first practical tungsten-filament light bulbs in 1904, dramatically outperforming earlier carbon-filament designs. In 1923, German scientists at Osram, including Franz Skaupy and K. Schröter, pioneered cemented carbide—also called hardmetal—by bonding tungsten carbide with cobalt. Although the original patent expired, Friedrich Krupp AG commercialized the technology extensively during World War II for cutting tools and armor-piercing applications.
- Throughout the 20th century, tungsten and tungsten alloys expanded into new roles. Although tungsten light bulbs gradually declined in popularity after 1950 due to evolving lighting technologies, the metal found renewed purpose in high-performance coatings introduced in 1969 for corrosion resistance. Today, tungsten remains essential in advanced sectors such as semiconductor fabrication, aerospace components, and precision electronics, continuing its legacy of enabling technological progress.
Applications of Tungsten
The combination of tungsten’s record-setting melting point and retained hardness at elevated temperatures positions it perfectly for extreme-duty environments. Common uses include light bulb filaments, cathode ray tubes, vacuum tube components, arc welding electrodes, gas tungsten arc welding (GTAW) systems, and electrical discharge machining (EDM) electrodes. These applications leverage tungsten’s ability to withstand intense heat and electrical loads without deformation or rapid wear.
Beyond electrical and thermal uses, tungsten serves a wide spectrum of industries including construction, aerospace engineering, mining operations, semiconductor manufacturing, consumer goods, jewelry design, laser welding systems, industrial machinery, electronics assembly, lighting technology, and healthcare equipment. Buyers researching tungsten solutions often ask practical questions such as “What tungsten applications work best for high-temperature tooling?” or “How is tungsten utilized in aerospace and defense components?” Exploring these real-world scenarios supports better matching of material grades to specific operational demands.
Design of Tungsten
Tungsten and its various alloys offer outstanding electrical conductivity and thermal management properties, making them highly valued in electrical engineering and power systems. Their strong resistance to corrosion further enhances suitability for harsh chemical or outdoor environments. Despite these strengths, pure tungsten tends to exhibit brittleness in its raw state, which can complicate forming and machining processes, particularly when components face mechanical stress or repeated thermal cycling. Engineers therefore frequently specify alloyed variants or specialized processing methods to improve ductility while preserving core performance characteristics.
When designing with tungsten, teams commonly evaluate factors such as operating temperature range, required hardness, electrical resistivity, and dimensional stability. Questions like “How should tungsten be specified for components exposed to thermal shock?” help guide material and processing decisions that optimize both functionality and manufacturability.
Tungsten Production Process
Manufacturing tungsten products typically begins with sintering or molding tungsten powder and selected alloys into dense billets or solid blocks. These starting forms are then transformed into finished shapes—including tungsten bar, tungsten sheet, tungsten plate, tungsten foil, rods, and tungsten wire—through precision techniques such as drawing, grinding, molding, and die cutting. Depending on the end use, additional finishing steps may involve chemical cleaning or precision grinding to achieve exact surface finishes and tolerances.
Available product conditions include black tungsten, which retains a protective lubricant and oxide layer from processing; cleaned tungsten, where the surface coating is chemically stripped; and ground tungsten, which undergoes diamond or silicon carbide machining for smooth, accurate dimensions and superior surface quality. Understanding these processing options helps buyers select the appropriate condition for their downstream fabrication or assembly requirements. Many procurement teams inquire: “What production steps ensure consistent quality in tungsten rod or sheet products?” or “How do different finishing processes affect performance in machining applications?”
Tungsten Images, Diagrams and Visual Concepts
 Tungsten is a durable tin white or steel gray metal, considered among the hardest metals found on earth.
Tungsten is a durable tin white or steel gray metal, considered among the hardest metals found on earth.
 Tungsten is a naturally occurring element extracted from the minerals wolframite and scheelite.
Tungsten is a naturally occurring element extracted from the minerals wolframite and scheelite.
 Products manufactured from tungsten are used in applications ranging from electrodes to construction machinery.
Products manufactured from tungsten are used in applications ranging from electrodes to construction machinery.
 Cemented carbide, a common tungsten alloy, is formed by combining tungsten carbide with cobalt as a binding agent.
Cemented carbide, a common tungsten alloy, is formed by combining tungsten carbide with cobalt as a binding agent.
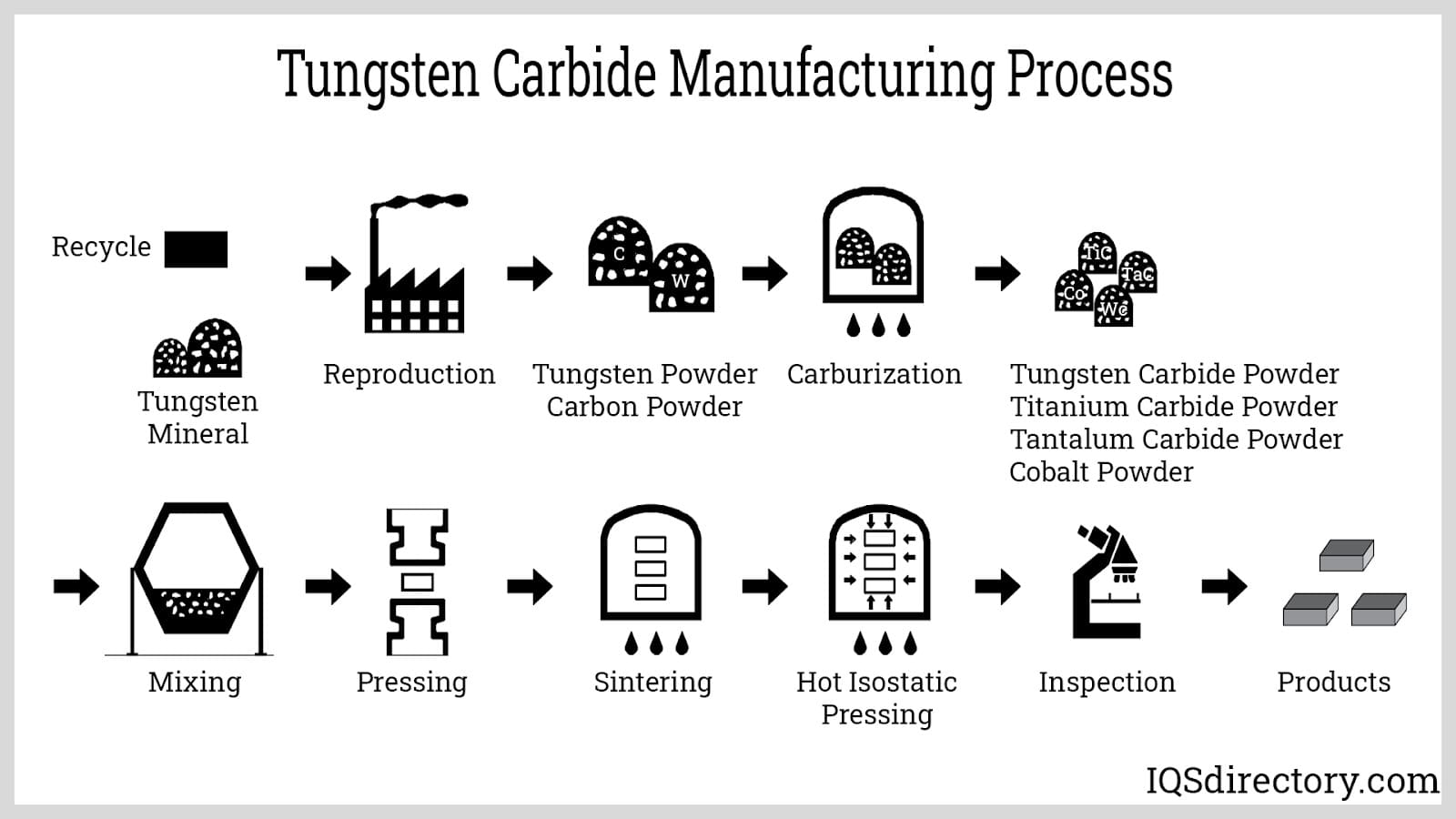 Tungsten carbide alloys are produced by reacting tungsten with carbon at high temperatures or through a low-temperature fluid bed process.
Tungsten carbide alloys are produced by reacting tungsten with carbon at high temperatures or through a low-temperature fluid bed process.
 Alloyed tungsten describes various alloys created by mixing tungsten with other metals.
Alloyed tungsten describes various alloys created by mixing tungsten with other metals.
 Heavy metal tungsten alloys contain up to 90% tungsten and may include around 2% thorium oxide.
Heavy metal tungsten alloys contain up to 90% tungsten and may include around 2% thorium oxide.
 Pure tungsten features high electrical conductivity and is used to make connections in circuit panels.
Pure tungsten features high electrical conductivity and is used to make connections in circuit panels.
Types of Tungsten
Leading tungsten suppliers categorize their offerings into five primary groups: tungsten carbide, cemented carbide, alloyed tungsten (including heavy metal tungsten alloys), pure tungsten, and tungsten-based chemicals. Within alloyed families, products are further differentiated by overall tungsten content and resulting tensile strength to match specific engineering needs.
- Tungsten Carbide
- Formed from equal atomic proportions of carbon and tungsten, this compound delivers exceptional hardness and wear resistance. Tungsten carbide surpasses the best grades of steel in hardness by a factor of two and exceeds the density of both titanium and conventional steel. Manufacturers produce over twenty distinct grades of tungsten carbide powder, each optimized for particular grain size, hardness, tensile strength, and thermal characteristics. Pressed and sintered tungsten carbide components serve extensively in metalworking, mining, and construction tooling, accounting for roughly 60 percent of tungsten carbide consumption in these sectors.
- Cemented Carbide
- Also referred to as hardmetal, cemented carbide bonds tungsten carbide particles with a cobalt matrix. The cobalt binder mitigates inherent brittleness, enabling the material to withstand high-pressure and impact loads while retaining superior wear properties. This combination makes cemented carbide a preferred choice for cutting tools, structural components, and heavy-duty mining or construction equipment.
- Alloyed Tungsten
- This broad family encompasses numerous formulations created by blending tungsten with complementary metals such as copper or iron. Common commercial examples include copper tungsten composites valued for electrical and thermal management applications. Suppliers tailor these alloys to balance conductivity, machinability, and mechanical performance across industrial and commercial uses.
- Heavy-metal Tungsten Alloys
- Containing a minimum of 90 percent tungsten, these dense alloys often incorporate small percentages of additional elements. Many formulations include approximately 2 percent thorium oxide to enhance performance in gas tungsten arc welding electrodes by improving electron emission and arc initiation stability. Their high density also suits applications requiring significant mass in compact volumes, such as ballast weights or radiation shielding.
- Pure Tungsten
- Characterized by outstanding electrical conductivity, pure tungsten finds primary use in electronics for creating reliable interconnections on circuit boards and other precision components where minimal resistivity is essential.
- Tungsten-based Chemicals
- The least common commercial form, these specialized compounds support production of organic dyes, phosphor pigments for displays, and phosphorescent materials used in medical x-ray intensifying screens.
Products Produced from Tungsten
Tungsten serves as the foundation for an extensive range of finished goods, including tungsten electrodes for welding and machining, incandescent and specialized lighting bulbs, drilling bits and mining tools, precision industrial machinery components, defense-related items, tungsten boats used in vacuum evaporation and thin-film coating processes, heavy construction equipment parts, and x-ray shielding screens. Tungsten alloys appear in armaments, heat sinks for electronics, turbine blades in power generation, dense counterweights and ballast materials, and durable industrial wear parts. Surface coatings of tungsten or tungsten carbide can also dramatically extend the operational lifespan of cutting tools and forming dies exposed to abrasive conditions.
Among the most prevalent tungsten products are welding electrodes. In electric arc welding processes, these electrodes deliver high-voltage current to workpiece surfaces, generating the intense localized heat needed to fuse metals. They play equally vital roles in electrical discharge machining and gas tungsten arc welding systems. Teams specifying tungsten components often investigate questions such as “Which tungsten products are best suited for EDM electrode applications?” or “How do tungsten boats improve vacuum coating uniformity?” These insights guide selection of the right form and alloy for each unique process requirement.
Things to Consider When Choosing Tungsten
Successful sourcing of tungsten products begins with partnering with a knowledgeable and reliable tungsten supplier capable of addressing project-specific needs. Key evaluation criteria include the supplier’s technical expertise, ability to meet budget targets, adherence to delivery schedules, and capacity to maintain consistent quality across order volumes. Confirming that the manufacturer understands exact material specifications, finishing requirements, and regulatory compliance further reduces risk and supports smooth project execution.
A practical approach involves reviewing detailed company profiles and websites, then contacting several shortlisted providers to discuss requirements directly. Comparing responses on lead times, customization capabilities, and support services helps identify the partner best aligned with both technical and commercial objectives. Many buyers ask: “What questions should I ask potential tungsten suppliers to ensure they can deliver the required quality and volume?” Structured supplier evaluation streamlines procurement and increases confidence in the final material selection.
Overseas Market of Tungsten
Global demand for tungsten remains robust due to its diverse applications in high-performance industries. Annual worldwide production totals approximately 45,000 tons, with the majority originating from mines in China and Russia. Other notable producers include Portugal, Austria, Peru, and Bolivia. Within the United States, domestic mining activity stays minimal, influenced by environmental considerations, competitive import pricing, and policy shifts. As a result, American manufacturers depend heavily on imported raw and semi-finished tungsten supplemented by extensive recycling programs that recover and reprocess the metal efficiently.
Collaborating with U.S.-based tungsten suppliers and fabricators offers distinct advantages, including streamlined communication, shorter lead times, reduced exposure to international shipping disruptions or tariffs, and full alignment with domestic regulatory standards. Even when overseas sources provide attractive base pricing, the overall value proposition of working with local partners—encompassing logistics simplicity and compliance certainty—frequently outweighs pure material cost differences. Recycling further bolsters supply chain resilience by providing a steady, environmentally responsible source of high-quality tungsten for new production cycles.
Tungsten Terms
- Alloy
- A solid solution or homogeneous mixture formed by combining two or more metals or elements.
- Brittle
- Prone to breaking under stress; brittle materials exhibit minimal deformation before fracturing.
- Filament
- A thin metal wire, typically made of tungsten, that emits light when electricity passes through it in a bulb.
- Melting Point
- The specific temperature at which a solid material transitions to a liquid state.
- Superalloy
- An alloy exhibiting high strength, hardness, and excellent resistance to corrosion, usually based on nickel, cobalt, or iron.
- Tensile Strength
- In engineering, the maximum stress a material can withstand before failure under tension.
- Wolfram
- Another name for tungsten, originating from wolframite ore, the principal source of the metal.